What Are Graphene Batteries?
Graphene batteries are energy storage devices that incorporate graphene — a single-atom-thick sheet of sp²-bonded carbon — into one or both electrodes to enhance electrochemical performance. Rather than replacing conventional lithium-ion chemistry outright, most graphene battery technologies use graphene or its derivatives (graphene oxide, reduced graphene oxide, graphene nanoplatelets) as additives, coatings, or structural components within existing electrode architectures. The result is improved rate capability, cycle life, energy density, and thermal management compared to conventional carbon-only electrodes.
The term “graphene battery” is used loosely in both research literature and marketing materials. In rigorous usage it encompasses: lithium-ion cells with graphene-modified anodes or cathodes, lithium-sulfur cells with graphene sulfur-host frameworks, sodium-ion cells using hard carbon–graphene composite anodes, lithium–air cells with graphene air electrodes, and hybrid supercapacitor–battery devices exploiting the double-layer capacitance of graphene alongside faradaic battery reactions.
Why Graphene Improves Battery Electrodes
The performance advantages of graphene in battery electrodes stem directly from its exceptional physical properties:
- Electrical conductivity: Graphene’s in-plane electron mobility (~200,000 cm²/V·s for pristine monolayer) creates a highly conductive network within electrode films, reducing ohmic losses during fast charge and discharge.
- High surface area: Theoretical surface area of ~2,630 m²/g for single-layer graphene provides abundant active sites for lithium ion intercalation and double-layer charge storage.
- Mechanical flexibility: Graphene’s tensile strength (~130 GPa) and elastic modulus (~1 TPa) allow it to accommodate volume changes during cycling without fracturing — a critical advantage for silicon and sulfur electrodes that expand 300–400% during lithiation.
- Chemical versatility: Functional groups on graphene oxide (–OH, –COOH, epoxide) can anchor sulfur, metal oxides, and other active materials through covalent or electrostatic interactions, preventing aggregation and loss of active material.
- Thermal conductivity: High in-plane thermal conductivity (~5,000 W/m·K for suspended single-layer) helps distribute heat within electrode stacks, reducing thermal gradients that accelerate degradation.
Graphene in Lithium-Ion Battery Anodes
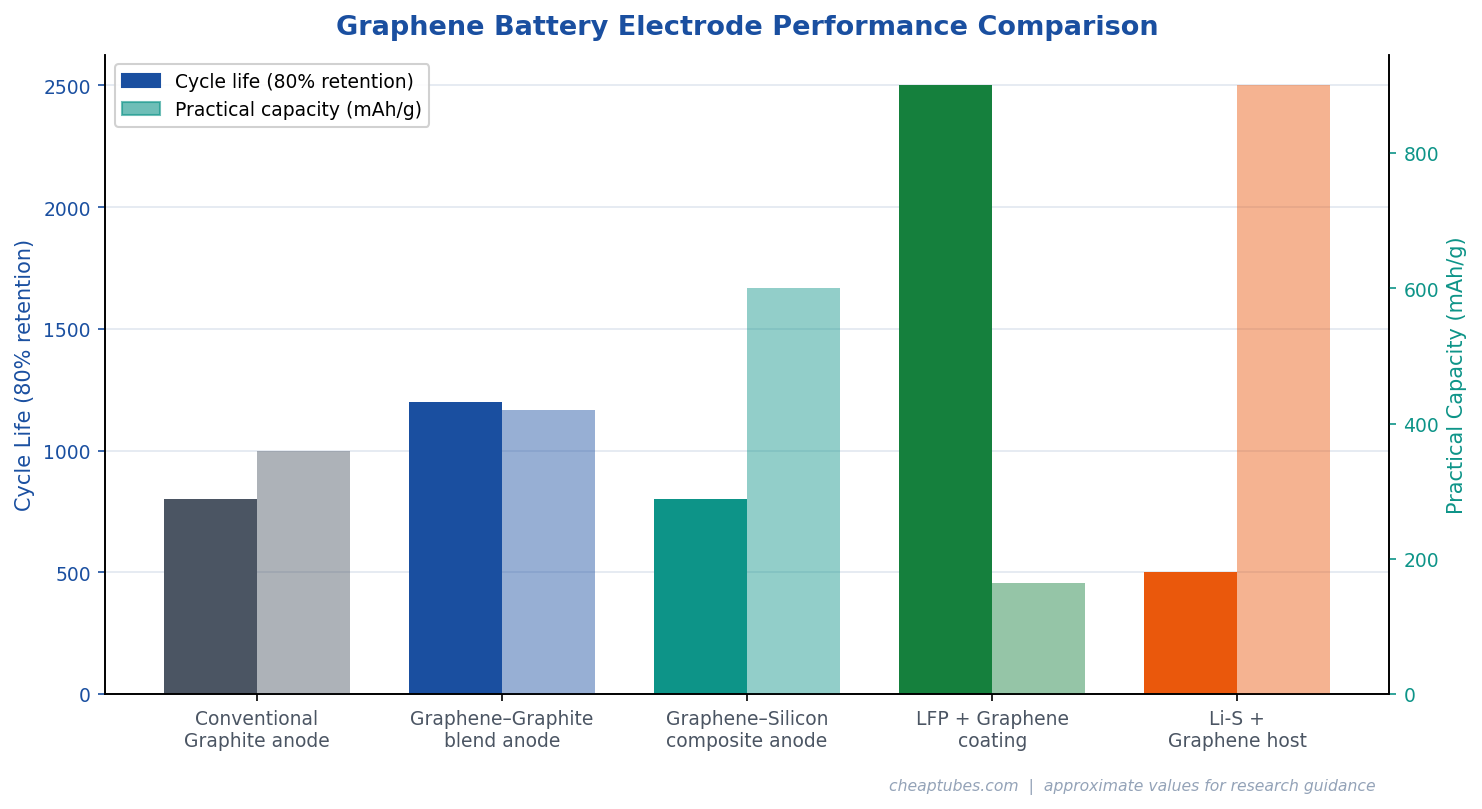
The anode of a conventional lithium-ion battery uses graphite, which stores lithium at a theoretical capacity of 372 mAh/g. Graphene-based anodes have demonstrated capacities of 700–1,100 mAh/g in research settings, though practical reversible capacities in full cells are typically 400–600 mAh/g due to solid-electrolyte interface (SEI) formation on high-surface-area graphene consuming lithium irreversibly on first charge.
The most commercially significant graphene anode application is not replacing graphite but enhancing silicon-graphite composites. Silicon offers ~3,580 mAh/g theoretical capacity but experiences catastrophic volume expansion (~300%) during lithiation, causing pulverization and rapid capacity fade. Wrapping silicon nanoparticles in graphene sheets or embedding them in reduced graphene oxide (rGO) scaffolds creates a flexible buffer that accommodates expansion while maintaining electrical contact throughout thousands of cycles. Several major cell manufacturers, including Panasonic, Samsung SDI, and emerging startups, have incorporated graphene-silicon composite anodes into next-generation 21700 and 46xx cylindrical cells.
Graphene in Lithium-Ion Battery Cathodes
Cathode materials including LFP (LiFePO₄), NMC (LiNiMnCoO₂), and NCA (LiNiCoAlO₂) have inherently low electronic conductivity that limits rate capability at high charge/discharge currents. Coating these particles with graphene nanoplatelets or conductive carbon derived from graphene oxide creates percolating conductive networks that dramatically reduce internal resistance and enable faster charge rates without sacrificing energy density.
LFP–graphene composite cathodes have become particularly important for EV and grid storage applications because LFP is lower-cost, thermally safer, and longer-lived than NMC, but was historically limited by poor rate performance. Graphene-coated LFP cathodes have enabled C-rates of 10C and above — meaning full charge in 6 minutes — while maintaining 90%+ capacity after 2,000+ cycles, making them competitive with NMC at fast-charge applications.
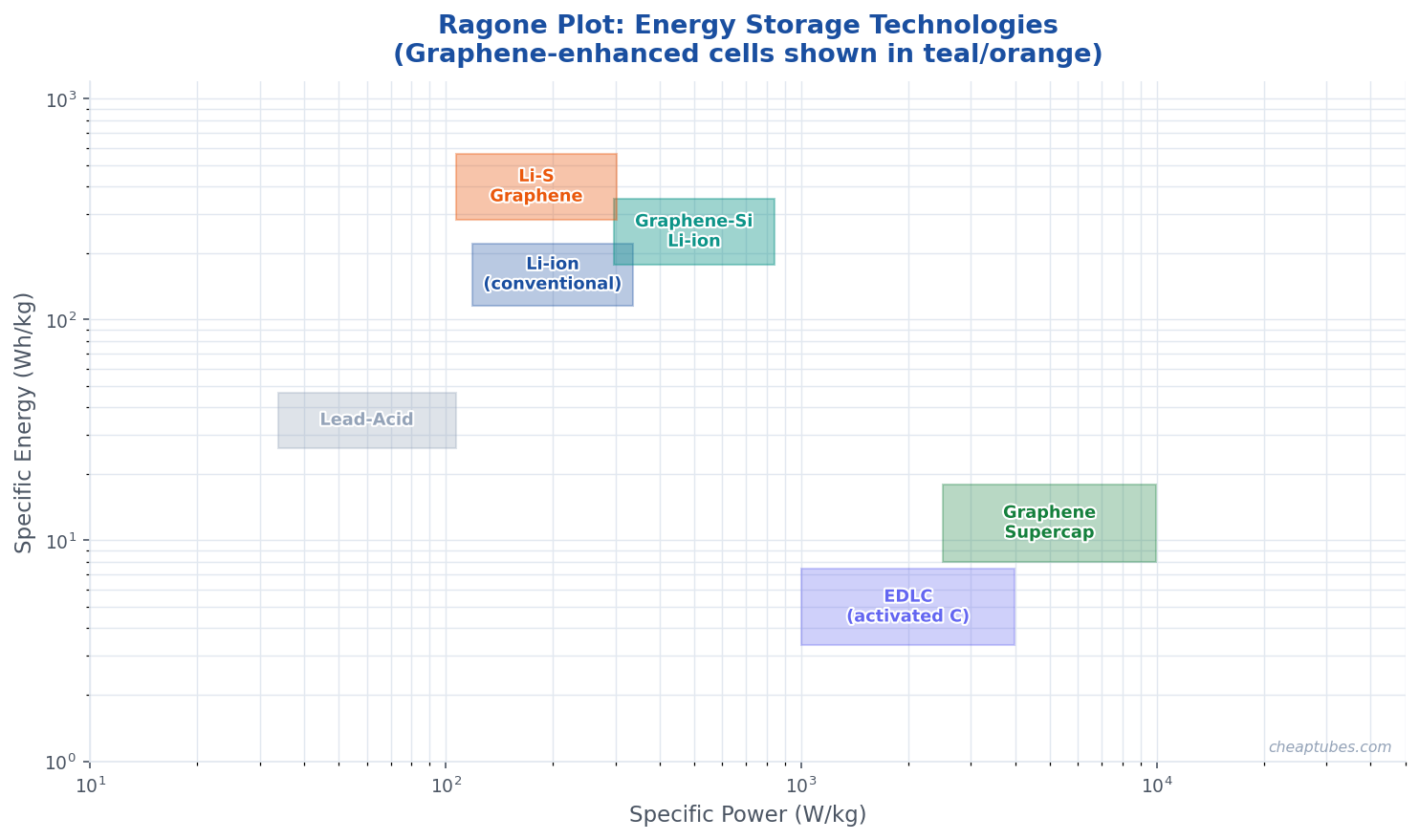
Graphene in Lithium–Sulfur Batteries
Lithium–sulfur (Li-S) batteries offer a theoretical energy density of ~2,600 Wh/kg, roughly 5× higher than current lithium-ion technology, making them a leading candidate for long-range aviation and grid storage. However, the polysulfide shuttle — where soluble intermediate discharge products (Li₂S₄ to Li₂S₈) dissolve into the electrolyte and migrate to the lithium anode — causes rapid self-discharge and capacity fade that has historically blocked commercialization.
Graphene and graphene oxide play several roles in addressing these challenges. Three-dimensional rGO aerogels serve as conductive sulfur hosts that physically confine polysulfides within the cathode structure. Nitrogen-doped graphene provides chemical anchoring sites (pyridinic N, pyrrolic N) that bind polysulfide anions through Lewis acid–base interactions, reducing dissolution. Graphene interlayers placed between the cathode and separator act as polysulfide barriers while providing additional conductive surface area. These approaches have extended Li-S cycle life from tens of cycles to several hundred, bringing practical application closer.
Graphene in Sodium-Ion Batteries
Sodium-ion batteries (SIBs) are emerging as a cost-competitive alternative to lithium-ion for stationary grid storage because sodium is far more abundant and geographically distributed than lithium. Sodium ions (ionic radius 1.02 Å) are too large to intercalate efficiently into conventional graphite, so SIBs use hard carbon — disordered, turbostratic carbon — as the anode material. Graphene nanoplatelets blended with hard carbon precursors, or reduced graphene oxide as a co-anode component, increase the interlayer spacing and defect density that accommodates sodium ions, improving both capacity and rate capability.
CATL and HiNa Battery Technology have both announced sodium-ion cells for EV and grid applications. As SIB anodes are optimized, graphene-derived carbons are expected to play an increasing role in achieving the 300+ Wh/kg targets that would make sodium-ion cells competitive with entry-level lithium-ion for mobility applications.
Graphene Supercapacitors and Hybrid Devices
Electrochemical double-layer capacitors (EDLCs, supercapacitors) store charge electrostatically at the electrode–electrolyte interface rather than through faradaic reactions, enabling extremely high power density (10–100 kW/kg) and millions of cycles with no chemical degradation. Activated carbon is the standard electrode material, but graphene’s higher theoretical surface area and better conductivity have driven intense research into graphene-based supercapacitors.
Practically, graphene sheets tend to restack during electrode fabrication, reducing accessible surface area back toward graphite levels. Strategies to maintain high surface area include: introducing spacer molecules or nanoparticles between layers, crumpling graphene sheets into a morphology that resists restacking, templating graphene onto 3D porous scaffolds, and using laser-scribed or microwave-exfoliated graphene that retains a porous structure. Graphene-based supercapacitors have achieved gravimetric capacitances of 150–300 F/g and energy densities of 70–85 Wh/kg — comparable to lead-acid batteries — while retaining the power density advantage of conventional supercapacitors.
Hybrid lithium-ion capacitors (LICs) combine a battery-type anode (often graphene-enhanced hard carbon or pre-lithiated graphite) with a capacitor-type cathode (activated carbon or graphene), targeting the middle ground between batteries and supercapacitors for applications like regenerative braking, UPS systems, and industrial power buffers.
Thermal Management in Battery Packs
Beyond electrode chemistry, graphene’s thermal conductivity makes it valuable as a battery pack thermal management material. Graphene-enhanced thermal interface materials (TIMs) placed between cells and cooling plates in EV battery packs reduce junction temperatures under fast charging, extending cycle life and enabling higher sustained charge rates. Graphene foils and papers produced by compressing graphene nanoplatelets achieve in-plane thermal conductivities of 700–1,500 W/m·K — several times higher than copper foil — at a fraction of the weight.
Graphene nanoplatelets with high aspect ratio and platelet diameter of 5–25 µm are well suited for thermal management applications, where heat must be spread laterally through the electrode stack or TIM layer. Products with surface areas of 120–750 m²/g cover the range from structural conductive additives to high-surface-area supercapacitor electrodes.
Current Commercial Landscape
As of 2025, fully graphene-based batteries remain primarily a research and development technology, while graphene as an additive or performance enhancer in conventional lithium-ion and emerging chemistries has entered commercial production. Notable developments include:
- Real Graphene (Canada): Markets USB-C power banks using graphene-enhanced lithium polymer cells, claiming 20-minute full charge and 1,500-cycle life for consumer electronics.
- Grabat (Spain): Demonstrated graphene polymer battery cells targeting 1,000 Wh/kg, though independent verification of claimed performance in production cells has been limited.
- Lyten (USA): Has raised substantial venture funding for its Li-S graphene battery platform, targeting EV applications with cells demonstrating 900+ Wh/kg at the material level.
- Samsung SDI, CATL, Panasonic: Incorporate graphene-silicon composite anodes and graphene conductive additives in commercial 21700 cells and next-generation prismatic formats without marketing them as “graphene batteries.”
Graphene Materials for Battery Research
Researchers developing graphene battery technologies require materials with well-defined and consistent properties. Key specifications include: number of layers (monolayer vs. few-layer vs. graphene nanoplatelets), lateral platelet size (affects aspect ratio and restacking tendency), oxygen content (C:O ratio for graphene oxide and rGO grades), surface area (BET, m²/g), and electrical conductivity (S/m for rGO and GNP powders).
Cheap Tubes supplies a complete range of graphene materials for battery electrode research, including graphene oxide for slurry-based electrode fabrication, reduced graphene oxide for conductive scaffolds and rGO aerogels, and graphene nanoplatelets for cathode conductive additives and thermal management films. All products are supplied with full analytical documentation including TEM, Raman, TGA, and BET data to support reproducible research.
Key Metrics for Evaluating Graphene Battery Claims
The graphene battery space attracts significant marketing hyperbole. When evaluating claims from suppliers, startups, or published papers, focus on specific metrics to distinguish genuine advances from promotional noise. Gravimetric energy density (Wh/kg) and volumetric energy density (Wh/L) should be reported at the full cell level, not just the active material level, since packaging, current collectors, separator, and electrolyte dilute material-level numbers by 30–60%. Cycle life should be specified at a defined capacity retention threshold — for example, 80% capacity after N cycles at a defined C-rate under defined temperature conditions. First-cycle coulombic efficiency matters for graphene anodes: high-surface-area graphene materials often have low first-cycle efficiency due to SEI formation, requiring pre-lithiation or excess cathode capacity to compensate.
Rate capability curves — capacity plotted versus C-rate from C/10 through 10C — reveal how much energy is accessible at real-world charge rates versus only at slow laboratory rates. Thermal performance data, including heat generation per cycle at high C-rates and maximum sustained operating temperature, become increasingly important as fast-charging enters the mainstream for EVs and portable electronics. Asking for independent third-party validation or peer-reviewed publication data is a reasonable standard for any material claiming performance well beyond incumbent technologies. Cross-referencing supplier data sheets against published electrochemical literature allows researchers to set realistic expectations before committing to a specific graphene grade for electrode development.
Contact our technical team to identify the right graphene grade for your battery chemistry, or to request samples for preliminary electrode trials.
