Flexiphene Surfactant Sheets
Quick Summary: Flexiphene® Surfactant Sheets
- What it is — A patented amphiphilic graphene-based surfactant sheet that disperses carbon nanomaterials without contamination
- How it works — Hydrophobic graphene core wraps nanotube sidewalls; hydrophilic edges provide electrostatic stabilization (−70 mV zeta potential)
- Compatible materials — SWCNTs, DWCNTs, MWCNTs, graphene nanoplatelets, carbon black
- Key advantage — No surfactant residue to remove; Flexiphene becomes part of the final material
- Available as — Dry sheets, pre-dispersed solutions, and specialty conductive papers
Flexiphene® Surfactant Sheets
Dispersing carbon nanotubes in water, NMP, or a polymer matrix without contaminating your end product is one of the hardest practical problems in nanomaterials processing. Conventional surfactants work — until they don’t, because removing them damages the tubes or interferes with downstream chemistry. Flexiphene is a different approach entirely.
Flexiphene is a patented amphiphilic surfactant sheet (US Patent 10,049,783) developed by CTI Materials and available exclusively through Cheap Tubes. The hydrophobic graphene core adsorbs to nanotube sidewalls via π–π stacking; the hydrophilic edges stabilize the dispersion electrostatically — achieving −70 mV zeta potential in DI water with no small-molecule surfactant contamination to remove. Compatible with SWCNTs, DWCNTs, MWCNTs, graphene nanoplatelets, and carbon black in aqueous and organic systems.
Available as dry surfactant sheets, dispersed solutions, and specialty CNT papers. Contact us for application support, technical data, and pricing.
Flexiphene Surfactant Sheets: Frequently Asked Questions
What is Flexiphene?
Flexiphene is a patented amphiphilic surfactant sheet made from a graphene-based material. It is designed specifically to disperse carbon nanomaterials — including single-walled, double-walled, and multi-walled carbon nanotubes, graphene nanoplatelets, and carbon black — in aqueous and organic media. Unlike conventional surfactants, Flexiphene does not leave contaminating residues because its graphene core integrates directly into the final carbon-based material or composite.
How does Flexiphene disperse carbon nanotubes?
Flexiphene works through an amphiphilic mechanism: the hydrophobic graphene core of the sheet adsorbs onto the sidewalls of carbon nanotubes through π–π stacking interactions, while the hydrophilic edges of the sheet extend into the surrounding medium and provide electrostatic stabilization. This achieves a zeta potential of approximately −70 mV in deionized water, indicating a highly stable colloidal dispersion. The result is individualized nanotubes without the need for aggressive sonication or chemical functionalization.
What makes Flexiphene different from conventional surfactants like SDS or Triton X-100?
Conventional surfactants like SDS (sodium dodecyl sulfate) or Triton X-100 must be removed after dispersion — a difficult, often incomplete process that leaves residues degrading the final material’s electrical and mechanical properties. Flexiphene eliminates this problem: its graphene-based structure is chemically compatible with the carbon nanomaterials it disperses, so it can remain in the final product (films, composites, electrodes) without acting as an insulating or contaminating impurity. Performance can also be tuned by adjusting pH, sheet size, and the degree of chemical reduction.
What applications is Flexiphene used in?
Flexiphene is used wherever stable carbon nanomaterial dispersions are required: (1) Conductive inks and films for printed electronics; (2) Battery and supercapacitor electrodes where pure carbon networks are essential; (3) All-carbon nanocomposites combining CNTs and graphene; (4) Filtration membranes with tunable pore structures; (5) Chemical and biological sensors requiring defect-free nanotube surfaces; (6) Emulsions for drug delivery and cosmetic formulations. Its no-residue characteristic is especially critical for electrochemical applications where surfactant contamination reduces active surface area.
What forms does Flexiphene come in?
Flexiphene is available in three primary product forms: dry sheets (for adding to your own solvent/nanomaterial system), pre-dispersed nanotube solutions (ready-to-use CNT dispersions stabilized with Flexiphene), and specialty conductive papers (freestanding flexible electrodes combining CNTs and Flexiphene). Custom concentrations, nanotube types, and sheet sizes are available for research and scale-up applications.
Frequently Asked Questions
Is Flexiphene a surfactant or part of the final material?
Flexiphene is an amphiphilic graphene-based dispersant that becomes part of the final material — it does not need to be removed after dispersion. The hydrophobic graphene core wraps the nanotube sidewall while hydrophilic edges provide electrostatic stabilization (~−70 mV zeta potential), so once your dispersion is incorporated into a polymer, ink, or paper, Flexiphene contributes its own electrical and mechanical properties rather than acting as inert contamination.
Which carbon nanomaterials does Flexiphene disperse?
Flexiphene effectively disperses single-walled carbon nanotubes (SWCNTs), double-walled CNTs (DWCNTs), multi-walled CNTs (MWCNTs), graphene nanoplatelets, and carbon black. It is compatible with both aqueous and selected organic solvent systems — the aqueous emulsion grade for water-based processing, the solvent-based emulsion for NMP/DMF/IPA systems.
How much Flexiphene do I need per gram of CNT?
Typical loadings are 0.1–1.0 g Flexiphene per gram of CNT depending on tube purity, length, and target dispersion concentration. Higher Flexiphene-to-CNT ratios produce more stable dispersions at higher CNT concentrations. The Technical Data Sheet for each Flexiphene grade includes recommended starting ratios for SWCNTs, MWCNTs, and graphene nanoplatelets.
What’s the difference between dry sheets, aqueous emulsions, and solvent-based emulsions?
Dry sheets are the raw Flexiphene material for users who want to formulate their own dispersions. Aqueous emulsions are pre-mixed water-based concentrates ready to add to your CNT or graphene system. Solvent-based emulsions are pre-mixed in NMP, DMF, or other polar organic solvents for non-aqueous processing. Pre-mixed grades save dispersion development time; dry sheets give maximum formulation flexibility.
Is Flexiphene patented?
Yes. Flexiphene is covered by US Patent 10,049,783 (“Amphiphilic graphene-based surfactant and method for preparing nano material colloids”) and is supplied exclusively through Cheap Tubes by license from CTI Materials.
Showing all 4 results
-

Flexiphene Aqueous Nanomaterial Emulsions
View Flexiphene Aqueous Nanomaterial Emulsions -

Flexiphene Solvent Based Nanomaterial Emulsions
View Flexiphene Solvent Based Nanomaterial Emulsions -

Flexiphene Surfactant Solutions
View Flexiphene Surfactant Solutions -

Flexiphene SWCNTs Paper
$300.00 Add to cart
What Makes Flexiphene Unique?
Flexiphene’s key differentiator is its patented, tunable surfactant nanosheets. Standard surfactants are small molecules — Flexiphene is a two-dimensional graphene-based sheet with both hydrophilic edges and hydrophobic basal plane regions. This size-dependent amphiphilicity allows Flexiphene to stabilize interfaces that small-molecule surfactants cannot, and the stability and performance of the dispersion can be tuned by adjusting three parameters: solution pH, the size of the surfactant sheet, and the degree of chemical reduction.
The result is a surfactant that provides more uniform dispersions and more precise control over formulations than conventional small-molecule surfactants — without introducing metallic or polymer contamination into the final product.
<!– UPLOAD: flexiphene-dispersion-mechanism.png → replace with:
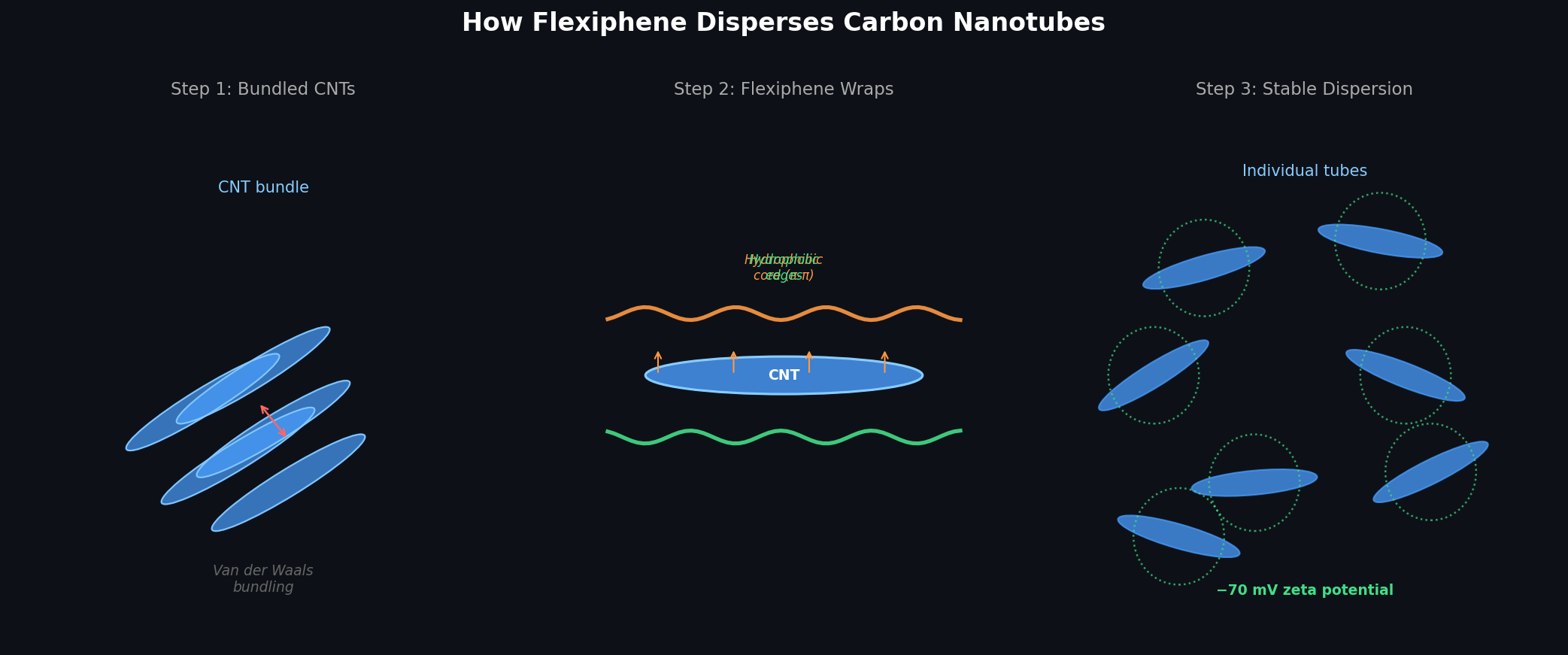
–>
How Flexiphene Works
| Property | Detail |
|---|---|
| Base Material | Graphene oxide (functionalized graphene nanosheet) |
| Amphiphilicity | Hydrophilic edges / hydrophobic basal plane |
| Tuning Parameters | pH, sheet size, degree of reduction |
| Compatible Nanomaterials | CNTs, graphene nanoplatelets, nanowires, nanoparticles |
| Compatible Media | Water, organic solvents, polymer matrices |
| Patent | US 10,049,783 (second patent pending) |
| Supplier | CTI Materials / Cheap Tubes (exclusive) |
<!– UPLOAD: flexiphene-stability-comparison.png → replace with:
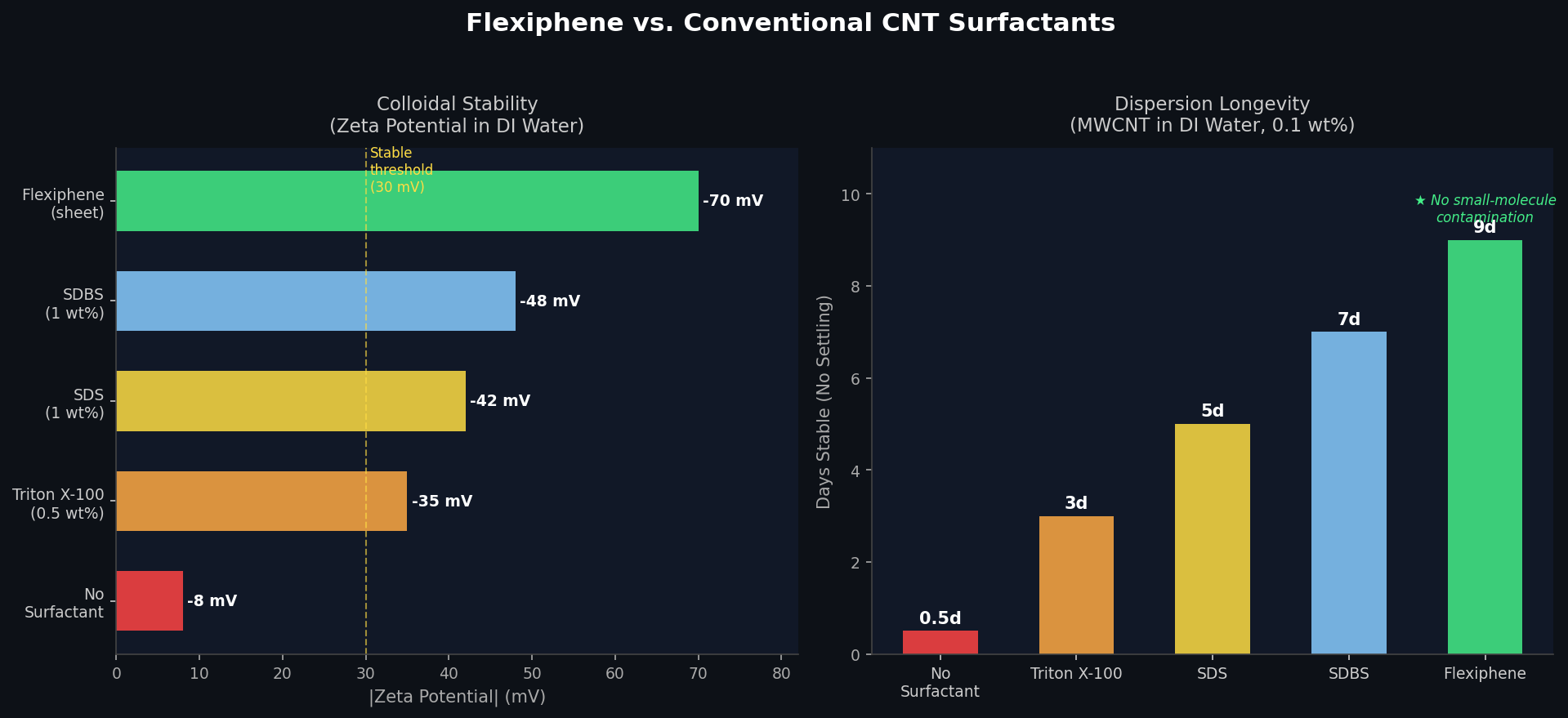
–>
Flexiphene Applications
Nanomaterial Dispersions
The primary application for Flexiphene is achieving stable, uniform dispersions of difficult-to-disperse nanomaterials. Carbon nanotubes have extremely high surface energy and tend to form tight bundles that resist separation even under aggressive sonication. Flexiphene sheets adsorb onto CNT surfaces and prevent re-agglomeration through steric and electrostatic stabilization — enabling stable aqueous and solvent dispersions at concentrations that would be impractical with small-molecule surfactants.
Emulsions
Flexiphene functions as a Pickering emulsifier — stabilizing oil/water interfaces using solid particles rather than molecular surfactants. Because Flexiphene sheets are large enough to irreversibly adsorb to the interface, Flexiphene-stabilized emulsions are significantly more stable than those produced with conventional surfactants. This is especially useful for polymer-based CNT or graphene formulations where long-term emulsion stability is required.
Conductive Films and Electrodes
Upon thermal or chemical reduction, Flexiphene converts from an insulating graphene oxide to a conductive reduced graphene oxide — while retaining its surfactant function. This means Flexiphene-dispersed CNT films can be made conductive without a separate post-processing step to remove the surfactant (which is required with conventional surfactants like SDS or Triton X-100, and which typically damages the CNT network). The resulting all-carbon conductive films have high conductivity and surface area suitable for electrode applications.
All-Carbon Nanocomposites
Combining CNTs with Flexiphene produces all-carbon composite structures where the graphene-based surfactant sheet becomes a functional matrix component rather than a processing aid to be removed. These CNT/graphene nanocomposites overcome re-agglomeration challenges, offering high electrical conductivity and large surface area for energy storage, EMI shielding, and structural reinforcement applications — without the laborious preparation methods required for other hybrid nanomaterial systems.
Membranes and Filters
Flexiphene’s ability to form ordered liquid crystal phases at high concentration allows it to be processed into oriented membranes with controllable pore structure. CNT/Flexiphene composite membranes show selective transport properties useful for gas separation, water purification, and ion exchange applications.
Sensors
The tunability of Flexiphene’s reduction state — from insulating to conductive — makes Flexiphene-based films responsive to chemical and electrochemical stimuli. Functionalized Flexiphene/CNT composites have demonstrated sensitivity to gases, pH, and specific analytes in electrochemical biosensor configurations.
<!– UPLOAD: flexiphene-applications-diagram.png → replace with:
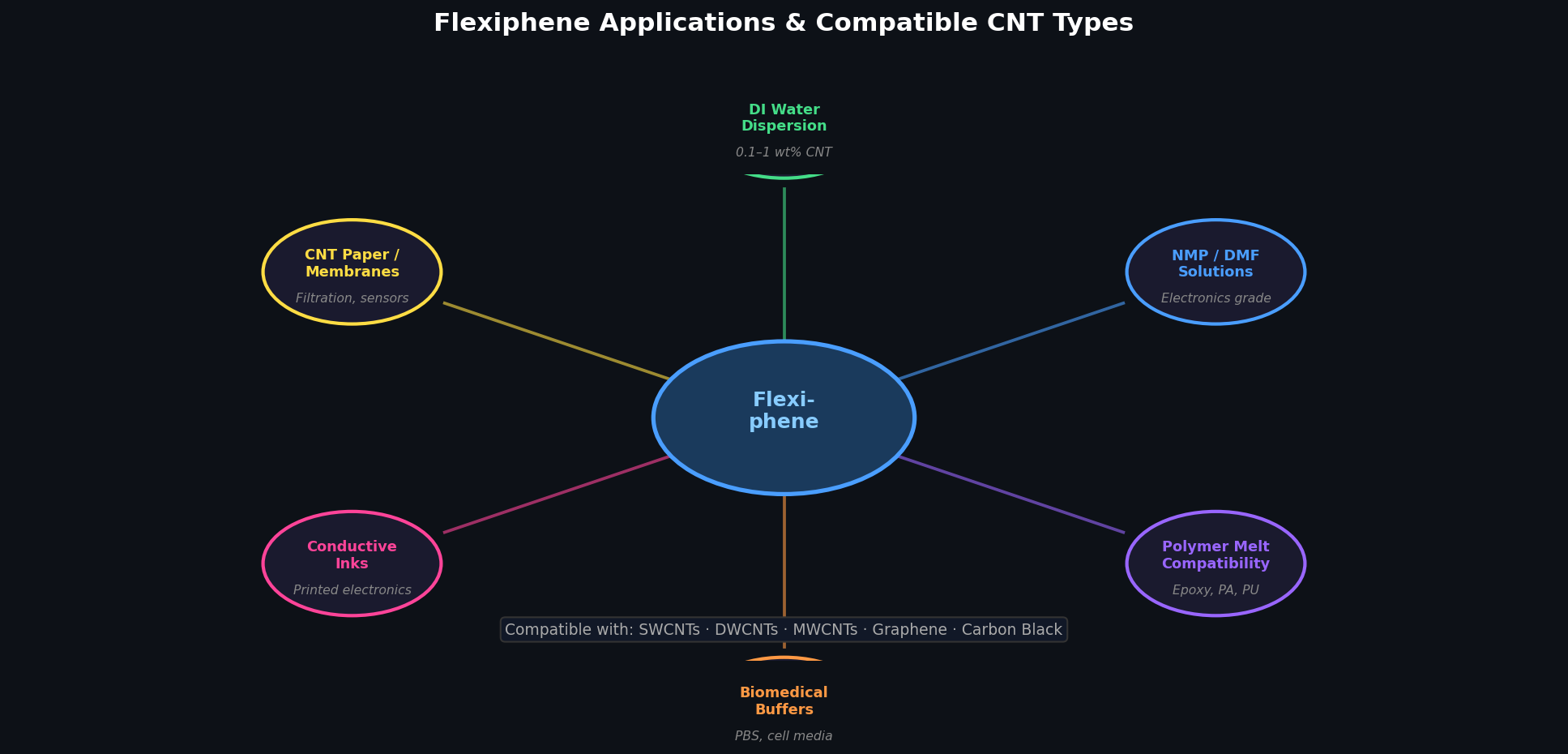
–>
Flexiphene Product Forms
Cheap Tubes offers Flexiphene in four forms to match different application workflows. Surfactant solutions are the base product — Flexiphene dissolved in aqueous or solvent medium, ready to use as a dispersant. Aqueous nanomaterial emulsions are pre-made dispersions of CNTs or graphene in water using Flexiphene as the stabilizer, delivered ready to use. Solvent-based nanomaterial emulsions serve the same purpose in organic solvent systems. Flexiphene SWCNTs paper is a freestanding all-carbon film of single-walled CNTs stabilized with Flexiphene, useful as an electrode or current collector in energy storage devices.
For technical questions, application development support, or custom formulations, contact us or book a call to discuss your specific requirements. Flexiphene is exclusive to Cheap Tubes.
Showing all 4 results
