Graphene Oxide
Quick Summary: Graphene Oxide (GO)
- What it is — oxidized graphene sheets bearing epoxy, hydroxyl, and carboxyl groups — water-dispersible without surfactants
- Why it matters — printable, biocompatible, and the most processable graphene precursor; reducible to rGO for conductive applications
- Compared to pristine graphene — lower native conductivity, but solution-processable, scalable, and chemically tunable
- Key applications — membranes, biosensors, drug delivery, printable inks (after rGO reduction), polymer composites, EMI films
- Available forms — single-layer GO, few-layer (2–4L), reduced GO (rGO), GO gel, freeze-dried GO — pricing from $125/g
- Choosing the right grade — see the Graphene Oxide Buying Guide for layer count and flake-size selection
Graphene Oxide
Finding consistent, well-characterized graphene oxide is harder than it should be. Batch-to-batch variation kills reproducibility. Cheap Tubes has supplied graphene oxide since 2007 — with full SDS documentation, transparent pricing, and responsive technical support.
Single layer, few layer, freeze-dried, and reduced GO grades are in stock now. Every lot is characterized by XPS and Raman so you know the C:O ratio, layer number, and functional group density before it arrives in your lab.
Need help choosing? Read our Graphene Oxide Buying Guide — a decision tree covering GO vs rGO, single vs few-layer, and 450 nm to 50+ µm flake size selection by application.
From $125/g. Ships from Vermont. Contact us if you need a custom specification or volume pricing.
Frequently Asked Questions
What is the difference between graphene oxide and reduced graphene oxide?
Graphene oxide (GO) is sp²/sp³ mixed graphene with epoxy, hydroxyl, and carboxyl groups attached — water-dispersible without surfactants but with reduced electrical conductivity (10⁷–10⁹ Ω·cm). Reduced graphene oxide (rGO) is GO that has been chemically, thermally, or electrochemically reduced to remove most oxygen functionality, restoring much of the sp² lattice and electrical conductivity (10⁻¹–10⁹ Ω·cm). Choose GO for membranes and biomedical work; choose rGO for conductive coatings, electrodes, and EMI shielding.
How many layers should I order?
Single-layer GO (>95% monolayer) is required for transparent conductors, ultra-thin membranes, and any application sensitive to flake stacking. Few-layer GO (2–4 layers) is the workhorse for composites, coatings, and energy storage where you need surface area and dispersion stability without paying for monolayer purity. Multilayer GO is fine for bulk composite reinforcement and water-treatment applications.
How do I disperse graphene oxide in water?
GO disperses spontaneously in water at concentrations up to ~5 mg/mL with gentle stirring or 2–5 minutes of bath sonication. The carboxyl and hydroxyl groups make it inherently amphiphilic. For higher concentrations or organic solvents, use 5–15 minutes of probe sonication with cooling, or order GO already dispersed in water, IPA, DMF, or NMP at the concentration you need.
What documentation comes with each order?
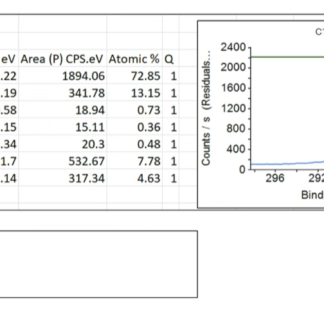
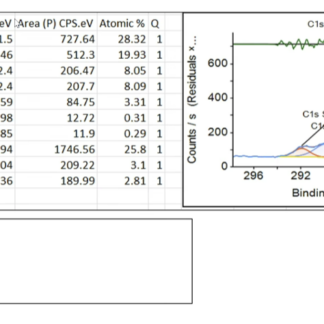
Every order includes a batch-specific Technical Data Sheet (TDS) with elemental analysis (C/O ratio), layer count, and lateral flake size, plus a current Safety Data Sheet (SDS), and a Certificate of Analysis (COA) on request. XPS, FTIR, Raman, AFM, and TGA characterization for the specific lot are available for research customers who need them for publication.
Can I get freeze-dried GO or GO gel?
Yes. Freeze-dried GO is offered as a free-flowing powder for users who want to formulate their own dispersion concentrations or store dry. GO gel (typically ~10–30 mg/mL) is offered for direct use without rehydration. Both are batch-traceable to the same source material as the standard GO dispersions.
What is the shelf life of graphene oxide?
GO dispersions are stable for 12+ months when stored at 4°C in the dark. Settling can occur with prolonged static storage and is reversed with gentle agitation or brief sonication — settling does not indicate degradation. Freeze-dried GO and GO gel can be stored at room temperature for 24+ months.
Where can I read more about choosing the right GO?
The Graphene Oxide Buying Guide covers form, layer count, flake size, reduction state (GO vs rGO), dispersion methods, and selection criteria organized by application.
Showing all 9 results
-
Exfoliated Reduced Graphene Oxide Powder
$115.00 Add to cart -
Few Layer Graphene Oxide 2-4L
$140.00 - $150.00 / per gram Select options -
Graphene Oxide Gel
$125.00 - $175.00 / per gram Select options -
Graphene Oxide Powder
$90.00 Add to cart -

Reduced Graphene Oxide
$190.00 - $200.00 / g Select options -
Reduced Graphene Oxide Industrial Grade
$110.00 Add to cart -
Single Layer Graphene Oxide
$140.00 - $150.00 / per gram Select options -

Single Layer Graphene Oxide 1-20um
$190.00 - $200.00 / per gram Select options -
Single Layer Graphene Oxide 450nm
$190.00 - $200.00 / per gram Select options
Choose the Right Grade
| Grade | Best For |
|---|---|
| Single Layer GO | Thin film research, membranes, biosensors |
| Few Layer GO (2-4L) | Composites, coatings, energy storage |
| Reduced GO (rGO) | Conductive films, electrodes, EMI shielding |
| GO Gel | Wet processing, filtration membranes |
| Freeze-Dried GO | Maximum solubility, long-term storage |
How to Order
- Select your grade from the products above
- Enter your quantity — price displays instantly
- Download SDS before you order if needed
Storage: Refrigerate at 2–8°C. Shelf life 12 months sealed.
Questions about which grade? Contact our technical team →
The Unique Properties of Graphene Oxide
Graphene oxide stands out owing to its unique properties like high surface area, functionality, and two-dimensional (2D) sheet-like structure. Its highly oxidized carbon atoms form a distinctive honeycomb hexagonal lattice pattern. The flakes range from nanometers to microns wide. Single-layer GO usually has a thickness of 0.7-1.2nm. The commonly available forms of GO include powder, solvent or polymer-dispersed form, or spin-coated film.
Graphene oxide can be easily dispersed in water, polymers, and solvents using ultrasonication or high shear methods. Although GO is electrically insulating, it can be transformed into a conductive substance by reducing it.
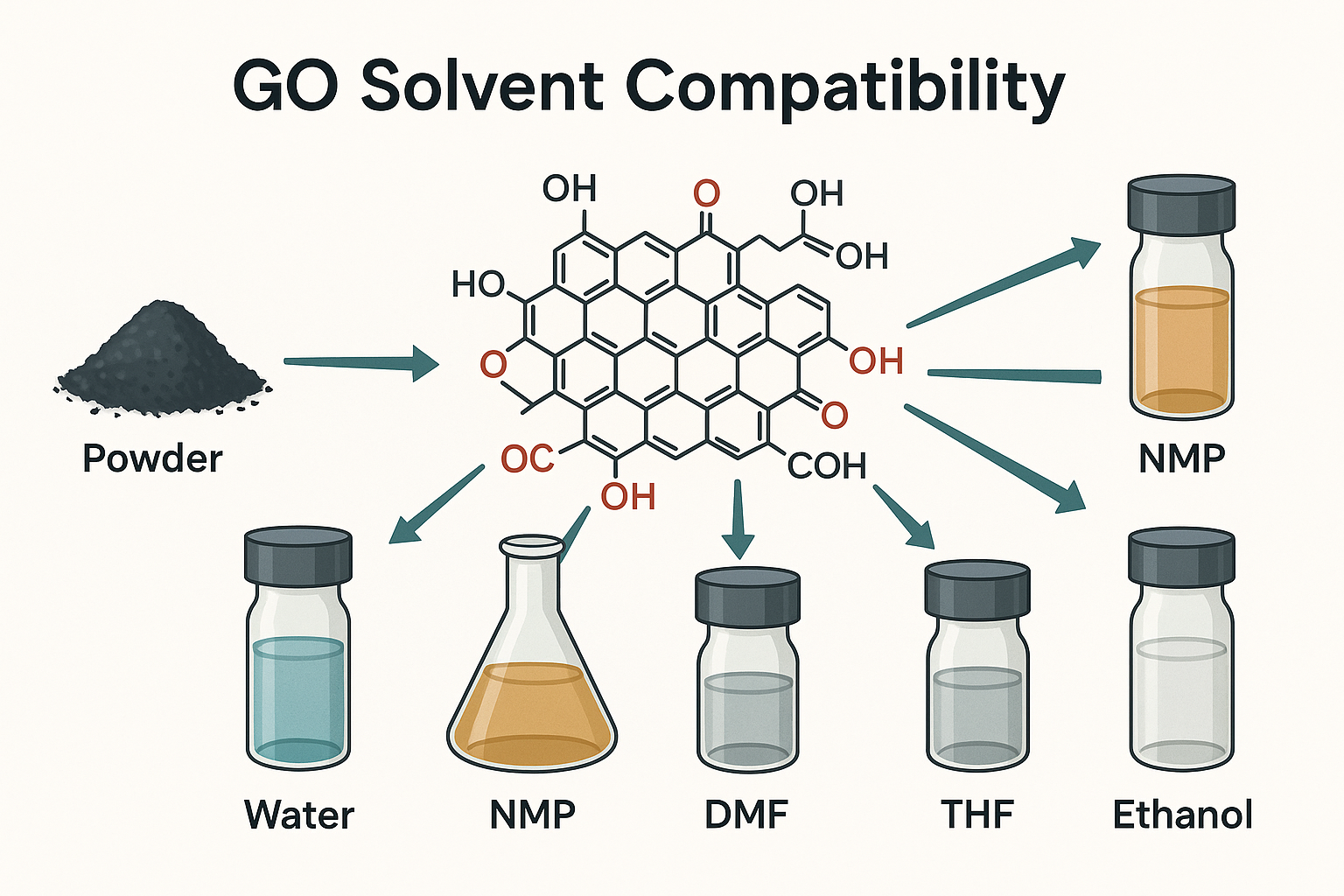
Understanding Graphene Oxide’s Physical Properties
GO usually possesses over 40% oxygen groups, including OH, COOH, and Epoxide groups, facilitating dispersion in Di water, NMP, DMF, THF, Ethanol, and other polar solvents. Fully oxidized, graphene oxide is a light brown (tan) solid powder with a C:O ratio between 2.1 and 2.9. Flake size usually ranges from hundreds of nanometers to tens of microns.
Electronic, Optical and Thermal Characteristics
GO conductivity can be improved through reduction. Optical transparency greater than 90% can be achieved with GO films less than 5 layers thick. The thermal conductivity of graphene oxide surpasses that of bulk graphite with similar interlayer spacing.
Graphene Oxide History
Initially known as graphite oxide, the story of graphene oxide spans over 150 years. Oxford chemist Benjamin Brodie first produced it in 1859. The safer Hummers method (1957) incorporated sulfuric acid, sodium nitrate, and potassium permanganate. Modern modifications such as the Tour method prioritize environmental and safety precautions.
Synthesis Method
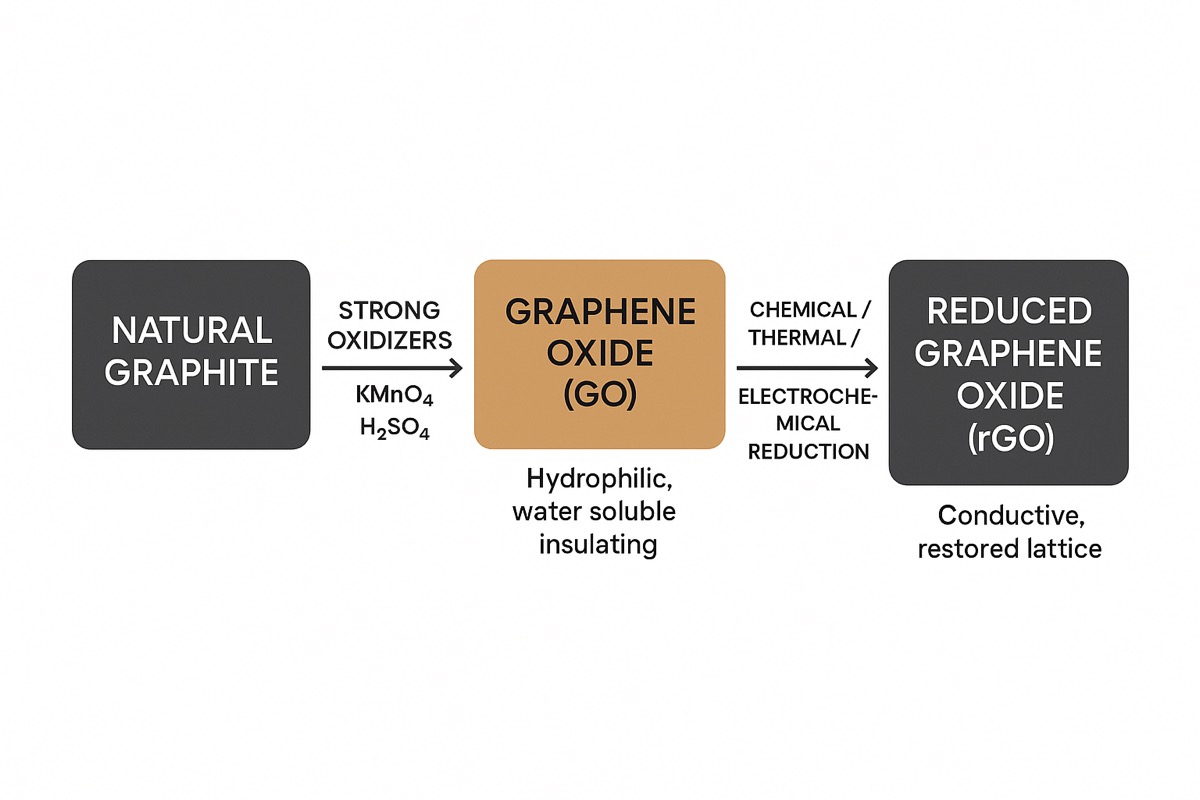
A modified Hummer’s method is used to make GO from graphite by treating it with strong oxidizers such as potassium permanganate, hydrogen peroxide, and sulphuric acid. This process exfoliates graphite into single or few-layer sheets and adds oxygen functional groups, making GO hydrophilic and water soluble.
Our GO is rinsed and centrifuged until the rinse water filtrate is pH neutral, then freeze dried to preserve solubility. Our GO is highly purified and flammability is not a concern.
Graphene Oxide Applications
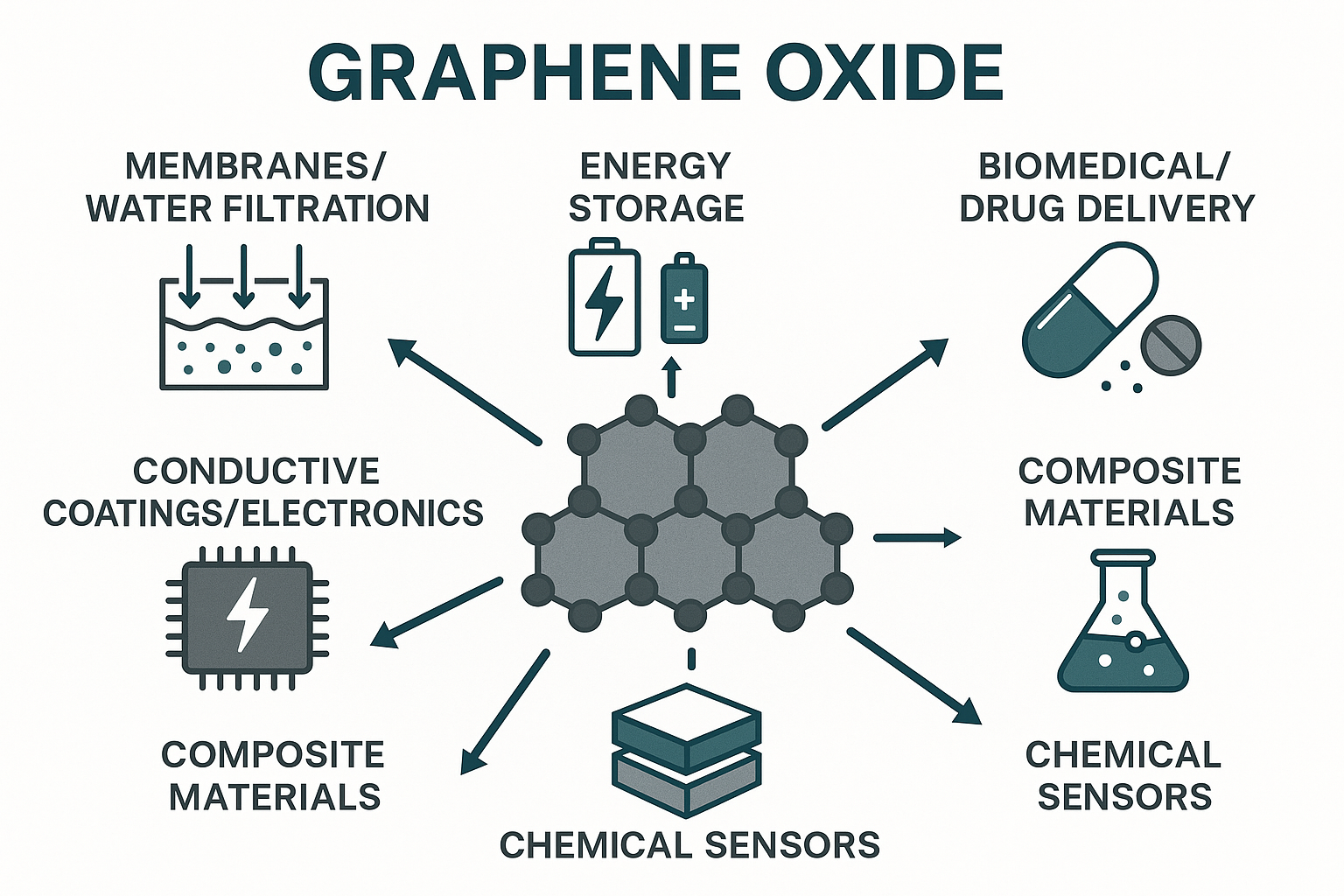
Graphene oxide applications include catalysis, drug delivery, solar cells, batteries, tissue scaffolding, water desalination and many other areas. Its high solubility and ability to be reduced enables solution processing. GO overcomes the well-known dispersion problems associated with carbon nanotubes.
Electronic Applications
Field effect transistors, chemical sensors, and biosensors have been fabricated using RGO. Transparent electrodes are important for LEDs, OLEDs, and solar cell devices.
Energy Storage Applications
RGO nanocomposites are used for high-capacity energy storage in lithium ion batteries and as electrode material in supercapacitors.
Biomedical Applications
GO’s biocompatibility enables drug delivery applications. Unlike CNTs, GO preparation avoids metal catalysts and metal impurities. Functional groups allow interaction with a wide range of organic and inorganic molecules.
Structure
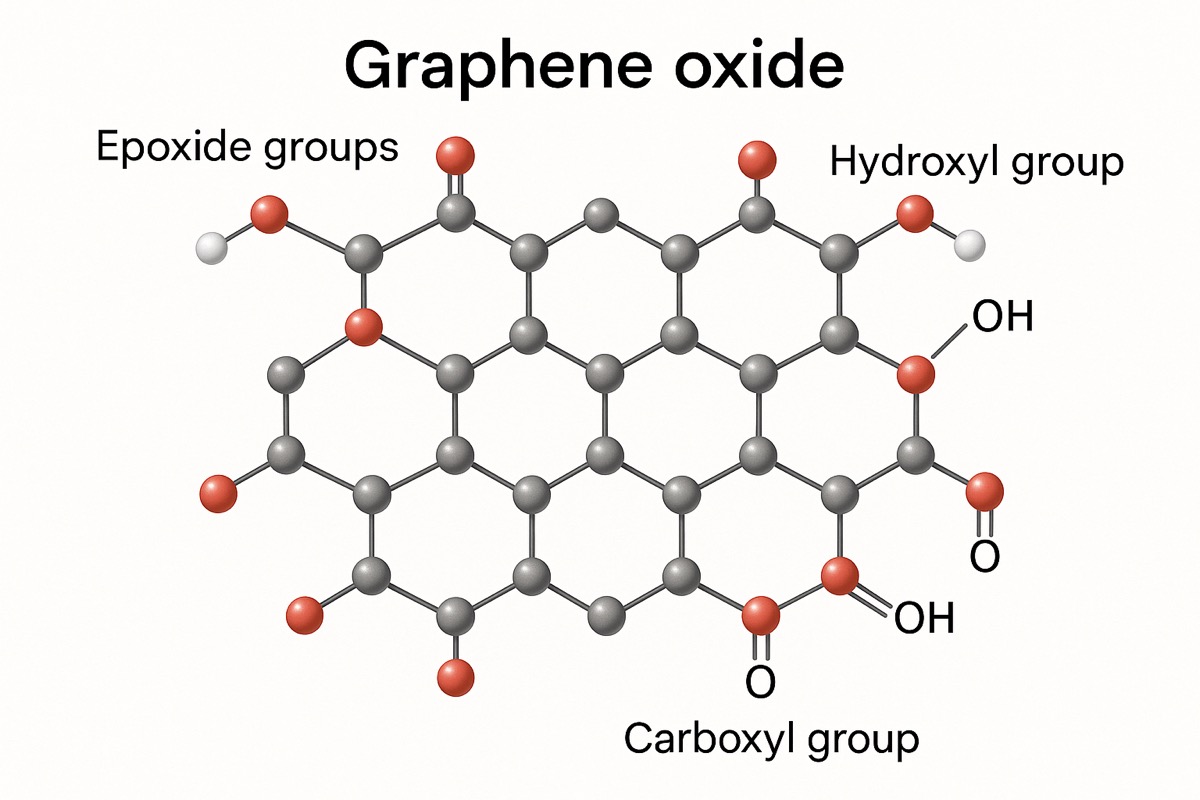
The 2D structure of graphene oxide features functional groups that give GO its legendary solubility. Surfactants are not needed when dispersing into polar solvents such as Di Water, NMP, DMF, THF, DCB, Ethanol, polymers, or others.
Molecular Weight
Graphene oxide chemical formula and molecular weight:
Chemical Formula: C140H42O20
Molecular Weight: 2043.856 g/mol
Reduced Graphene Oxide
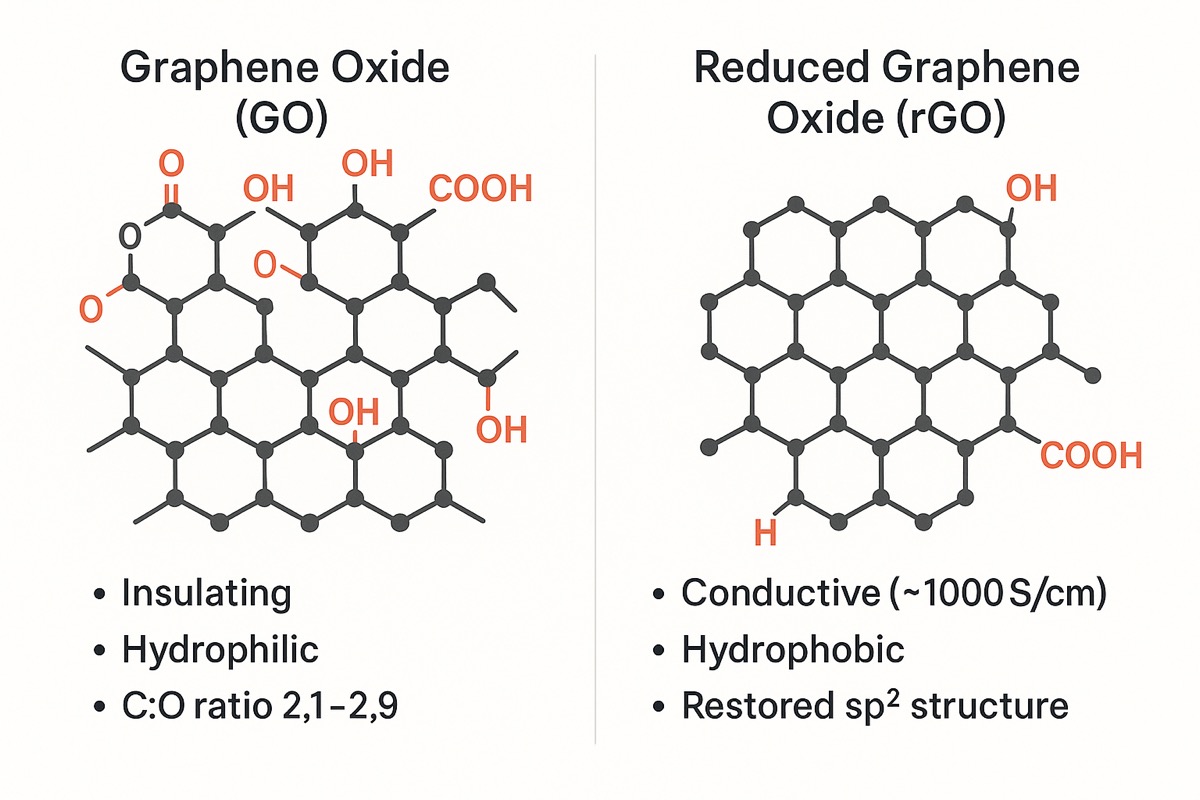
Reduced graphene oxide is synthesized as regular GO then reduced, removing surface functionality and restoring the molecular structure closer to pristine graphene. Common reduction methods include chemical (hydrazine), thermal (furnace, microwave), electrochemical, and laser/light-based approaches.
Graphene Oxide Price
Graphene oxide price is determined by production volumes and degree of purification. Most of our products are between $75–200/g. Our products are washed and centrifuged 15 times until filtrate is pH neutral, then freeze dried to preserve solubility. Not all suppliers take these steps. We recommend storing GO in a lab refrigerator; shelf life is approximately 6 months before functionality begins to decrease.
Difference Between Graphene & Graphene Oxide
2D sheets less than 10 layers thick are generally called graphene. The three main types are: CVD/epitaxial graphene films, graphene oxide (exfoliated via strong acid oxidation), and graphene nanoplatelets (plasma or chemically exfoliated).
Showing all 9 results
