Fullerenes
Quick Summary: Fullerenes
- What they are — closed-cage carbon allotropes (C₆₀, C₇₀, higher fullerenes) with discrete molecular structure and unique electronic and biological behavior
- Why they matter — strong electron acceptors, antioxidant properties, photoactive in organic photovoltaics, purifiable to molecular precision
- Compared to other nanocarbons — discrete molecules (not extended sheets or tubes), fully soluble in many organic solvents, chromatographically purifiable
- Key applications — organic photovoltaic devices, antioxidant cosmetics, MRI contrast agents, optical limiting, single-molecule electronics
- Available forms — C₆₀ >99%, C₇₀ >98%, C₆₀/C₇₀ mixtures, PCBM (PV-grade)
- Selection guide — see the Fullerenes: Properties and Applications guide
Fullerenes — C60 & C70
Whether you’re developing organic solar cells, characterizing electron transport through molecular junctions, or exploring fullerene photochemistry, your results depend on purity you can verify — not just a certificate of analysis number that came with the shipment.
Cheap Tubes has supplied HPLC-purified C60 (99%+) and C70 (99%+) to research labs since 2005, backed by representative UV-Vis and mass spec characterization data. From 100 mg for exploratory work to multi-gram quantities for scale-up. If our material doesn’t meet our published purity specifications, we’ll replace it or refund your order. Ships from Vermont.
C60 from $15/g, C70 from $30/g. Contact us for volume pricing or custom purity specifications.
Deeper reading: Fullerenes: Structure, Properties, and Applications — our research guide covering cage topology, synthesis, characterization, functionalization chemistry, and purity-grade selection.
Showing all 3 results
-
Carbon Fullerenes C60
$95.00 - $99.00 / per gram Select options -
Carbon Fullerenes C70
$225.00 - $250.00 / per gram Select options -

Fullerenes C60 99.9
$125.00 - $150.00 / per gram Select options
What Are Fullerenes?
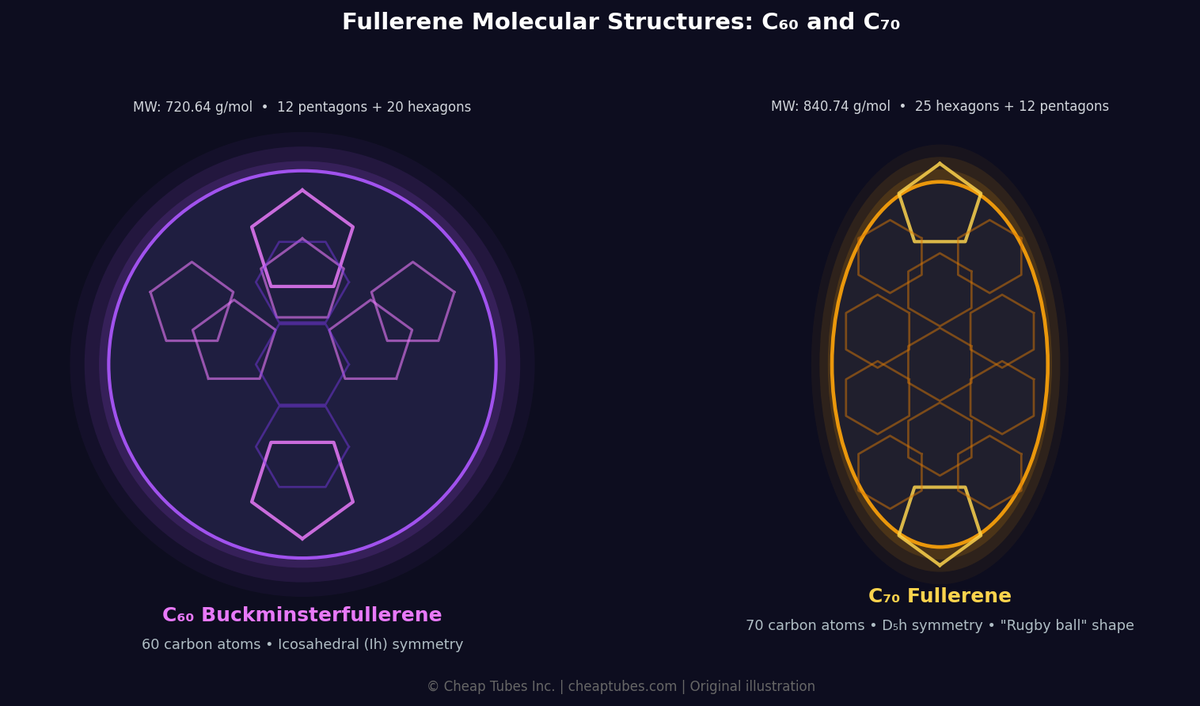
Fullerenes are carbon allotropes arranged in closed, hollow cage-like structures. The most well-known is Buckminsterfullerene (C60) — 60 carbon atoms in 20 hexagons and 12 pentagons, resembling a soccer ball at the nanoscale. Discovered in 1985 by Kroto, Curl, and Smalley (Nobel Prize, Chemistry, 1996). The family spans C60, C70, C76, C84, endohedral fullerenes, and functionalized derivatives.
C60 Properties
| Property | Value |
|---|---|
| Molecular Weight | 720.64 g/mol |
| Symmetry | Ih (icosahedral) |
| Cage Diameter | ~0.71 nm |
| Solubility in Toluene | ~2.8 mg/mL at 25°C |
| Solubility in CS2 | ~7.9 mg/mL at 25°C |
| Electron Affinity | 2.65 eV |
| HOMO-LUMO Gap | 1.9 eV |
| Max Li atoms per cage | 12 |
C60 is an excellent electron acceptor (triply degenerate LUMO, reversibly accepts up to 6 electrons). Soluble in aromatic solvents and CS2; insoluble in water in pristine form. Light-sensitive in solution — store protected from UV.
C70 vs C60
| Property | C60 | C70 |
|---|---|---|
| Molecular Weight | 720.64 g/mol | 840.74 g/mol |
| Shape | Spherical (Ih) | Ellipsoidal (D5h) |
| Visible absorption | Lower | Higher — broader spectrum |
| OPV performance | Baseline | 5–10% higher PCE |
| Cost | Lower | Higher (less abundant) |
Applications
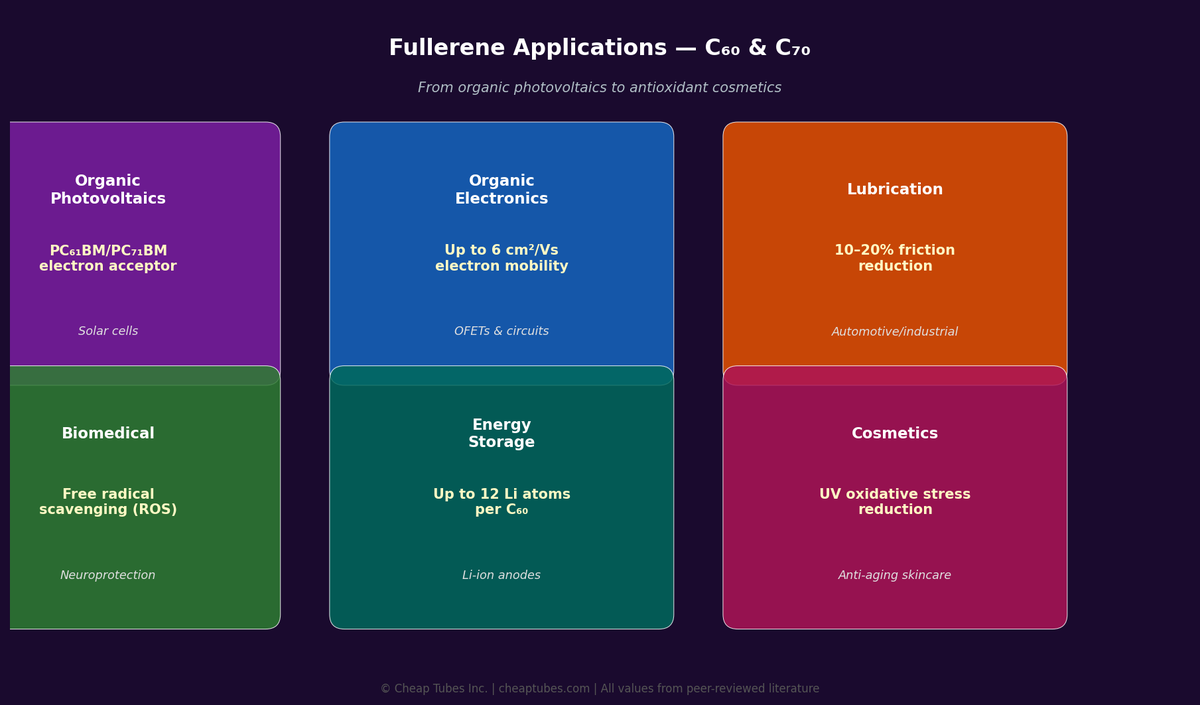
Organic Photovoltaics (OPV)
Fullerenes are the benchmark electron acceptor in bulk heterojunction organic solar cells. PC61BM and PC71BM (derivatives of C60 and C70) achieve PCEs of 8–11% in single-junction devices. C60 and C70 are direct precursors for these derivatives and are used in vacuum-deposited bilayer OPV architectures.
Organic Field-Effect Transistors
Thermally evaporated C60 films form n-type organic semiconductors with electron mobilities up to 6 cm²/Vs — among the highest for any organic n-type material. Used in flexible electronics, CMOS logic, and chemical sensors.
Lubrication and Tribology
C60’s spherical geometry enables it to function as a molecular ball bearing. Fullerene lubricant additives reduce friction coefficients by 10–30% in metal-on-metal contacts. Effective in high-vacuum and extreme-pressure environments where conventional lubricants fail.

Radical Scavenging and Antioxidant
C60 is arguably the most potent radical scavenger known — reacting with up to 34 methyl radicals per molecule. Its 3D pi system efficiently traps oxygen-centered and carbon-centered radicals. C60 in squalane is used in anti-aging skincare, outperforming vitamin C and E in peer-reviewed UV-oxidative stress studies.
Biomedical Research
Functionalized and water-soluble fullerene derivatives (fullerenols, amino-fullerenes) are being investigated for antiviral activity (HIV, SARS-CoV-2 protease), antibacterial coatings, photodynamic therapy (singlet oxygen generation), and neuroprotection in Parkinson’s and Alzheimer’s disease models.
Energy Storage
C60 can reversibly accommodate up to 12 Li atoms per molecule. Alkali-metal-doped fullerenes (K3C60) become superconductors at low temperature (Tc = 18K). Endohedral metallofullerenes are studied for quantum information applications.
Synthesis
Commercial fullerenes are produced by the Kratschmer-Huffman arc evaporation method: graphite electrodes are vaporized by high-current arc in helium atmosphere (~100 Torr). The resulting soot (75–85% C60, 10–15% C70) is extracted with toluene and purified to >99% by HPLC on a Buckyprep column. Identity confirmed by MALDI-TOF mass spectrometry and UV-Vis absorption.
Solubility and Handling
Prepare solutions by sonicating C60 in solvent for 15–30 minutes, then filter through 0.2 μm PTFE membrane. Solutions are photosensitive — store in the dark. Dry powder is stable indefinitely sealed, away from light and moisture. Handle with nitrile gloves; avoid inhalation of fine powder.
Grade Selection and Pricing
Cheap Tubes offers C60 (99%+, 99.5%+, 99.9%+) and C70 (99%+) with HPLC, MS, and UV-Vis characterization on every lot. Available from 100 mg to multi-gram quantities. C60 99% from $99/g; C70 99% from $250/g. Contact us for volume pricing and custom specifications.
Frequently Asked Questions
What is the difference between C60 and C70?
How do I dissolve fullerenes?
What purity grade should I order?
Are fullerenes air-stable?
What are typical applications of fullerenes?
Showing all 3 results
