Introduction To Graphene
Types Of Graphene
Properties of Graphene
Applications of Graphene
This Guide to Graphene Synthesis, Properties, and Applications is intended to convey a general understanding of these topics for both Scientists & Non-Scientists alike.
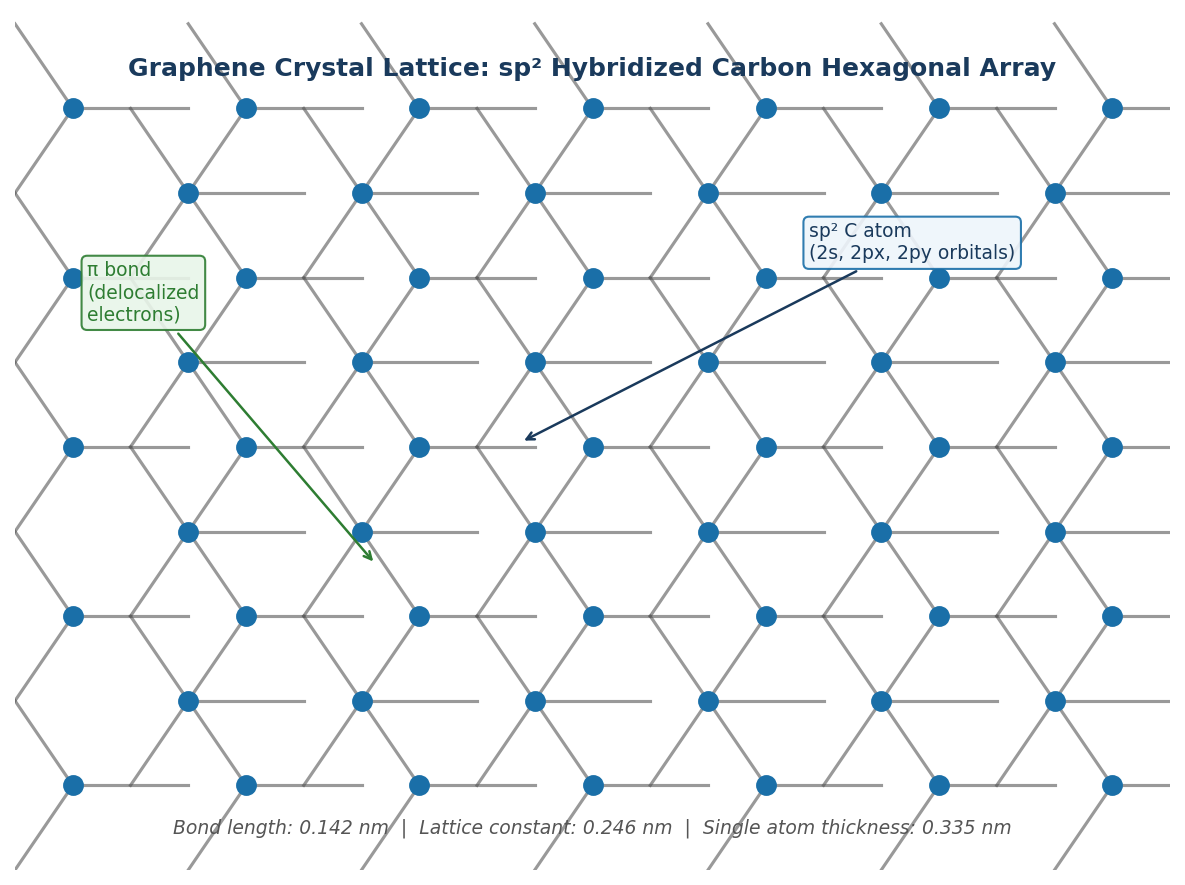
Introduction To Graphene
Types & Forms of Graphene
To gain the benefits of graphene oxide, it is typically dispersed, added into a formulation, made into a film or other nano-enabled product and then reduced to restore the graphene structure.
Reduced Graphene Oxide (rGO)
There are many methods to reduce graphene oxide (GO) into reduced graphene oxide (rGO), but most fall into three main categories: chemical reduction, thermal reduction and electrochemical reduction.
The other methods include hydrazine vapor treatment, annealing, laser and microwave reduction. The reduction process is vital to producing rGO, as it determines how consistent the rGO structure is with the GO precursor.
Many commercial producers of Graphene Nanoplatelets are in fact providing a product similar to industrial scale rGO as their GNP product. However this method differs from the rGO most people refer to which is a higher quality research product used for nano enabled devices.
Chemical reduction is a scalable method but can often result in poor yields and utilizes highly toxic materials such as hydrazine. rGO produced by this method generally exhibits a low surface area and has a low conductivity compared to the GO precursor.
Thermal reduction produces rGO with a high surface area that is close to the surface area of pristine graphene. However, the intense heating process causes a high-pressure build-up of carbon dioxide which causes structural damage to the graphene layers.
The structural imperfections can then give rise to a reduction in the overall mass (and yield against the theoretical output), vacancies, voids and it can hinder the mechanical strength of the material.
Electrochemical reduction shows the best results in terms of production and quality. The rGO produced is consistent with that of pristine graphene.
During the electrochemical process, the substrates (generally ITO or glass) are coated with a layer of GO and a current is passed through the material (via electrodes at either end of the substrate). rGO produced by this method have shown to have a high carbon to oxygen ratio and have exhibited conductivity comparable to that of silver.
The process also benefits from no toxic waste. This process does however suffer from issues regarding the feasibility of the scalability of the method.
Graphene Nanoplatelets (GNPs)
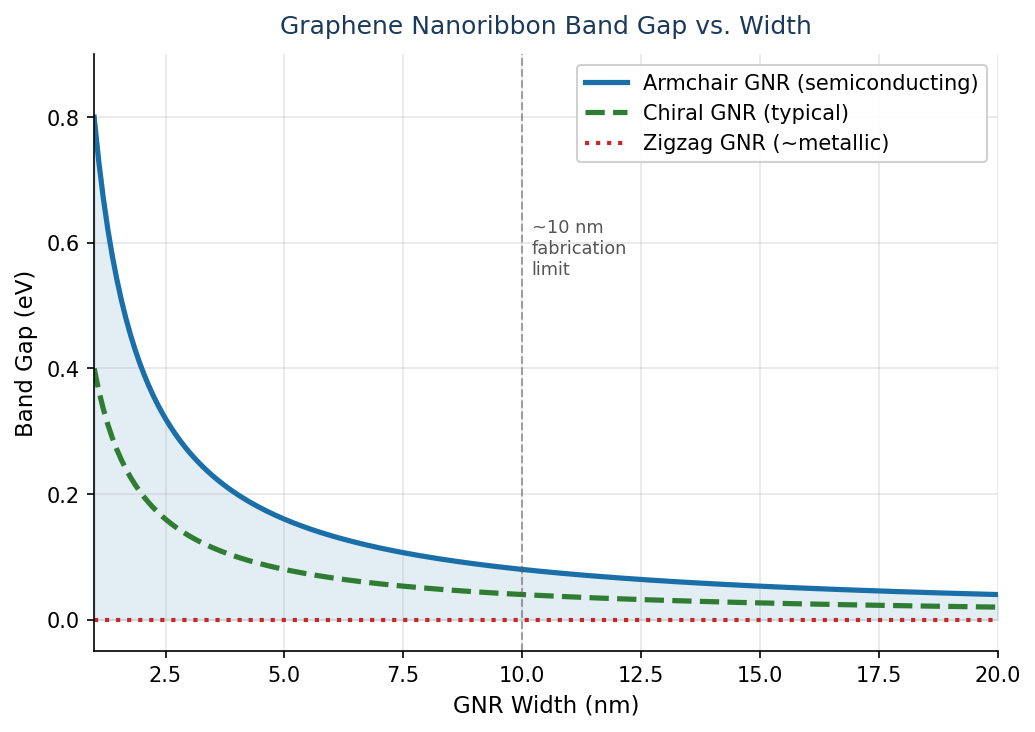
GNRs can enhance the performance of lithium-ion batteries through edge chirality effects. GNRs have a band gap that is inversely proportion to their width, which is dependent upon their edge chirality.
Chirality occurs at the edges because the electron confinement potential deforms the wave-function and causes the electrons move in a single direction, with more weight in the ‘positive x’ direction. This leads to a current in the ‘positive y’ direction.
For electronic applications, the edges in GNRs have shown the best results when armchair and metallic edges are present due to their semi-conducting abilities. Armchair edges also reduce the band gap energy when there is a defined width.
The energy at the edges of GNRs is proportional to their density and armchair edges are more tightly packed at the graphene interface, so the energy is higher than that of zig-zag edges. Such edges can be produced on GNRs by electron beam irradiation and electron beam lithography.
A GNR with a high concentration and purity of armchair edges has been found to provide highly efficient p-n junctions in electronic devices.
Graphene Aerogels
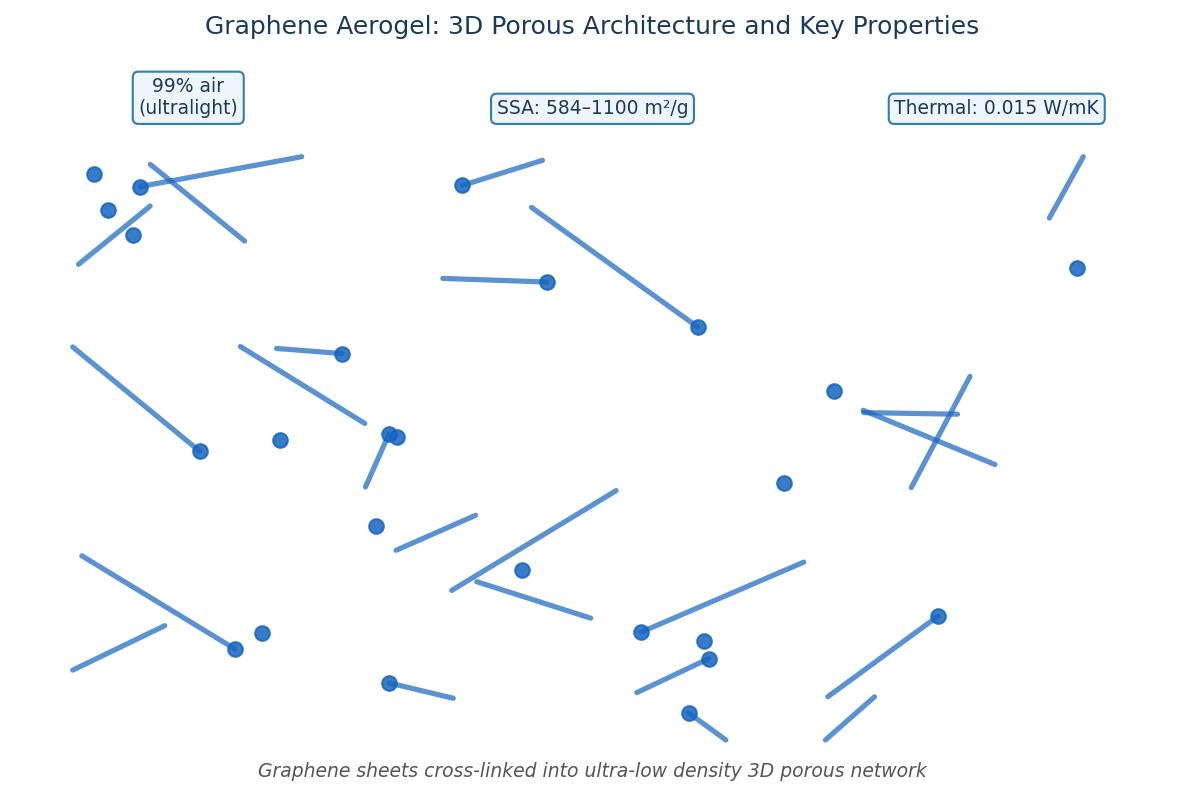
Carbon aerogels are derived by sol-gel synthesis methods and are a unique class of high-surface-area materials. Their high mass-specific surface area, electrical conductivity, environmental compatibility, and chemical inertness make them very promising materials for many energy related applications.
Recent developments in controlling their morphology make them especially well suited to super capacitor applications.
Aerogels are a special class of open-cell foams that exhibit many unique and interesting properties, such as low mass density, continuous porosity and high surface areas. These properties are derived from the aerogel microstructure, which consists of three-dimensional networks of interconnected nanometer-sized particles.
Aerogels are typically prepared by sol–gel methods, a process that transforms molecular precursors into highly cross-linked inorganic or organic gels that can then be dried using techniques such as supercritical drying, freeze drying, ect to preserve the insubstantial solid network.
For organic and carbon aerogels, the transformation involves the polymerization of multi-functional organic species into three-dimensional polymer networks.
Graphene Masterbatches
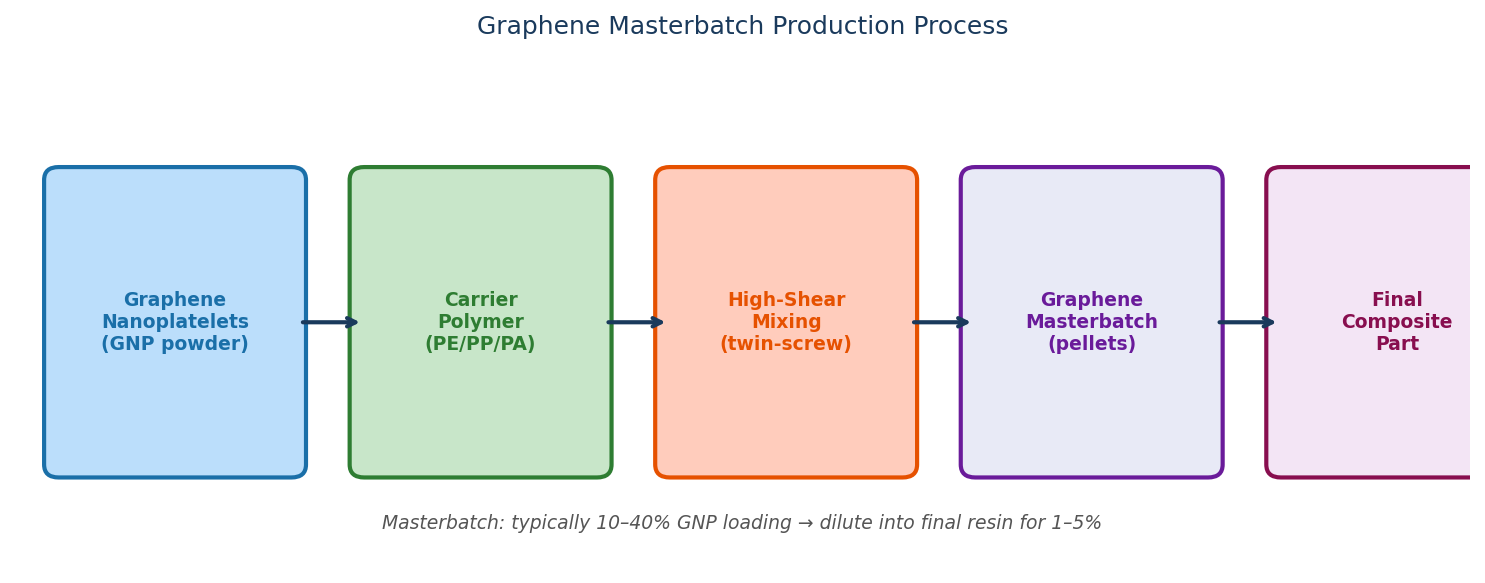
Graphene masterbatches are composite materials that contain a graphene-based compound (most commonly GO) and a polymer.
The graphene is used to enhance the properties of various common polymeric materials. Many polymers exhibit desirable properties such as low cost, low toxicity, bio-compatibility and chemical resistance, but they lack desirable mechanical properties.
By incorporating graphene nanoplatelets into polymer matrices, the polymers retain their original properties but benefit from enhanced rigidity and stiffness, while still being lightweight.
Using graphene as a filler compound rather than conventional inorganic materials can bring an enhanced electrical conductivity to the polymer, but it does have some issues.
In many graphene-based composites, graphene oxide acts as the dispersing support for other ions and molecules.
In polymer masterbatches, this can lead to problems as graphene doesn’t always disperse well in polymer phases (especially polyolefins) due to a lack of positive interactions at the grpahene polymer interface.
However, this can be overcome by the use of a surfactant, or by tailoring surface functionality of the graphene surface. The surfactant increases the surface interaction between the polymer and graphene.
If functionalized, the functional groups promote interaction between itself and the polymer molecules. If the functional groups aren’t compatible, you may observe what we call “islands of masterbatch” with easily observed islands of polymer in between well dispersed graphene-polymer masterbatches.
Properties Of Graphene
The properties of graphene are unique due to its all carbon structure and nanoscale geometry.
Electronic Properties
Because graphene has a delocalized pi-electron system across the entirety of its surface, the movement of electrons is very fluid.
The graphene system also exhibits no band gap, due to overlapped pi-electrons, allowing for an easy movement of electrons without the need to input energy into the system.
The electronic mobility of graphene is very high and the electrons act like photons, with respect to their movement capabilities.
The electrons are also able to move sub-micrometer distances without scattering. From tests done to date the electron mobility has found to be in excess of 15,000 cm2V-1s-1, with the potential of producing up to 200,000 cm2V-1s-1.
Thermal Properties
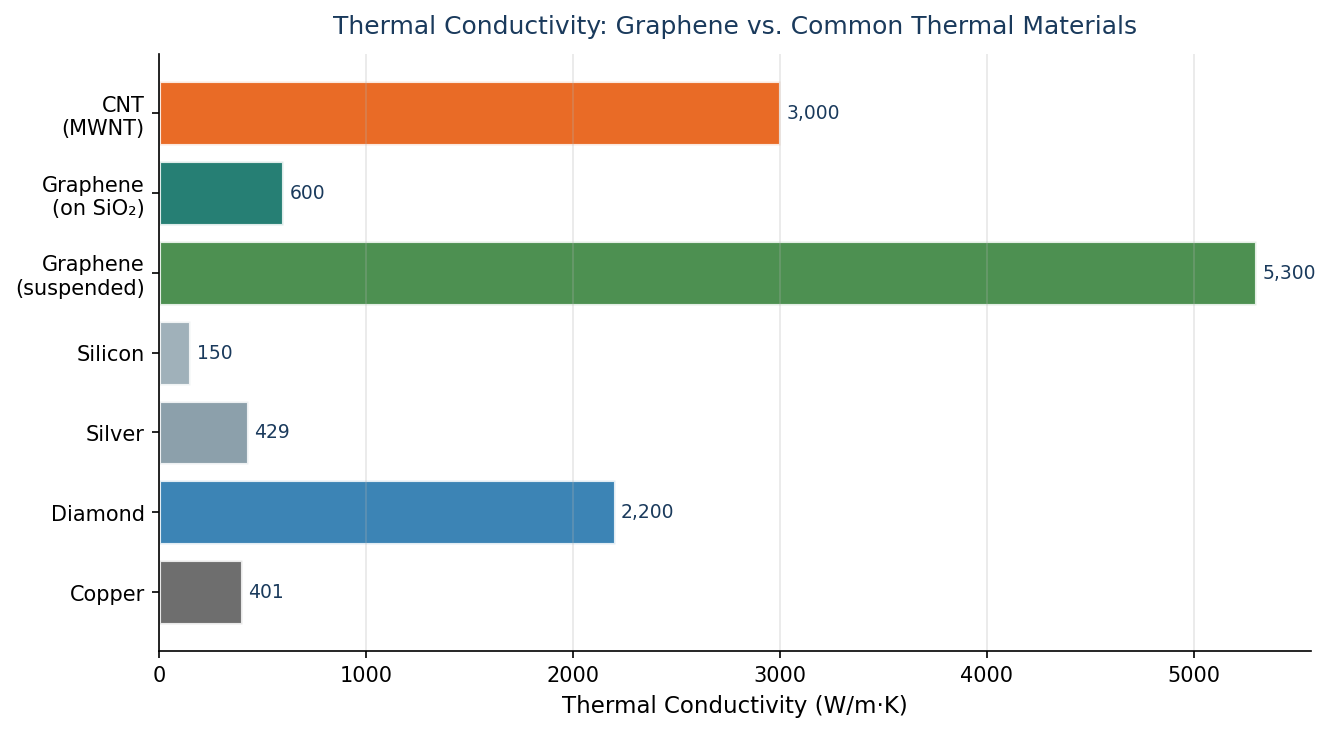
The repeating structure of graphene makes it an ideal material to conduct heat in plane. Interplane conductivity is problematic and typically other nanomaterials such as CNTs are added to boost interplane conductivity.
The regular structure allows the movement of phonons through the material without impediment at any point along the surface. Graphene can exhibit two types of thermal conductivity- in-plane and inter-plane.
The in-plane conductivity of a single-layered sheet is 3000-5000 W m-1 K-1, but the cross-plane conductivity can be as low as 6 W m-1 K-1, due to the weak inter-plane van der Waals forces.
The specific heat capacity for graphene has never been directly measured, but the specific heat of the electronic gas in graphene has been estimated to be around 2.6 μ J g-1 K-1 at 5 K.
Mechanical Strength
Graphene is one of the strongest materials ever discovered with a tensile strength of 1.3 x 1011 Pa. In addition to having an unrivaled strength, it is also very lightweight (0.77 mgm-2).
The mechanical strength of graphene is unmatched and as such can significantly enhance strength in many composite materials.
Flexibility/Elasticity
The repeating sp2 hybridized backbone of graphene molecules allow for flexibility, as there is rotation around some of the bonds, whilst still providing enough rigidity and stability that the molecule can withstand changes in conformation and support other ions.
This is a very desirable property as there are not many molecules that can be flexible and supportive at the same time. In terms of its elasticity, graphene has found to have a spring constant between 1-5 Nm-1, with a Young’s modulus of 0.5 TPa.
Applications of Graphene
There are many applications of graphene because it’s a revolutionary material. It has many applications replacing conventional materials as well as the ability to support applications previously not possible before the advent of 2D materials. The applications of Graphene are truly endless and many are yet to be conceived of yet.
Sensors
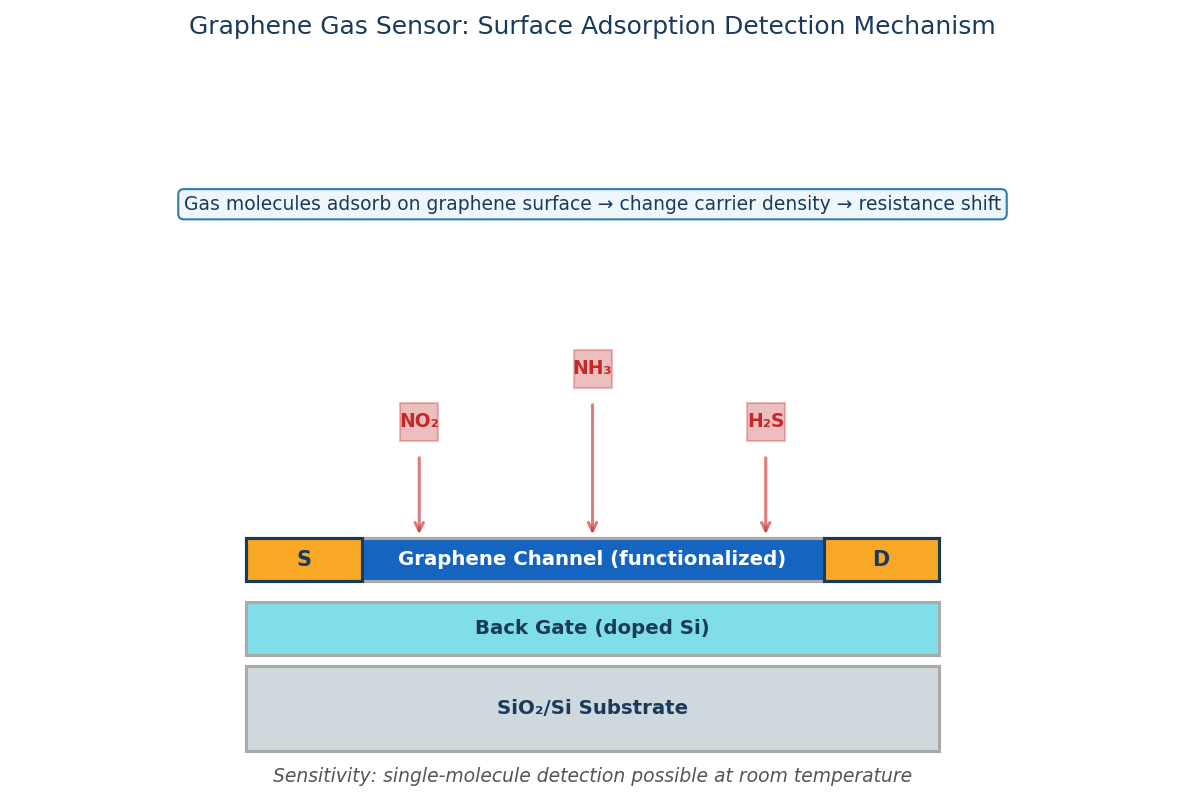
The ideal sensor is able to detect minute changes in its surrounding environment. Due to the planar and consitent arrangement of atoms in a graphene sheet, every atom within the sheet is exposed to the surrounding environment.
This allows graphene to effectively detect changes in its surroundings at micrometer dimensions, providing a high degree of sensitivity.
Graphene is also able to detect individual events on a molecular level. Many of graphenes properties are beneficial in sensor applications; as such, graphene could be used in sensors in various fields including bio-sensors, diagnostics, field effect transistors, DNA sensors and gas sensors, to name a few.
Batteries
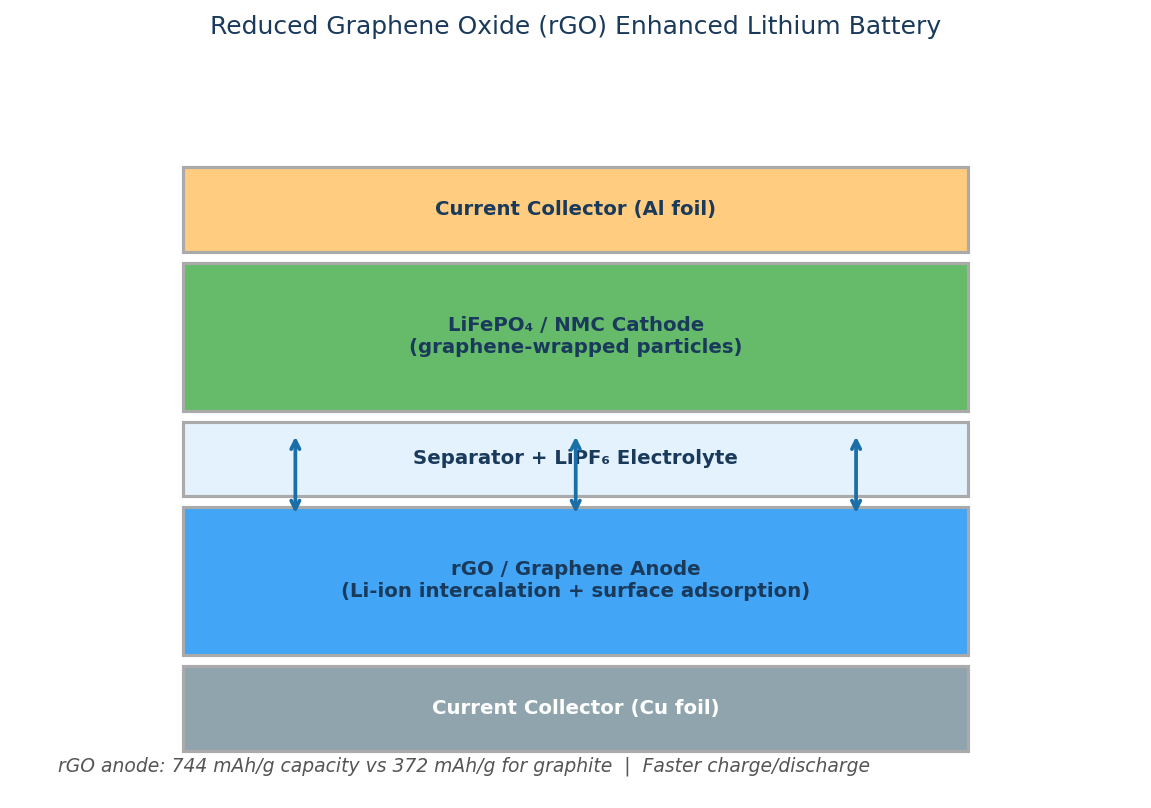
Graphene can be incorporated into both the anode or the cathode in various battery systems to increase the efficiency of the battery and improve the charge/discharge cycle rate.
The excellent electrical conductivity, surface area and dispersibility of graphene enhances the beneficial properties present in many traditional inorganic-based electrodes, whilst simultaneously relieving the electrodes of their limitations.
Due to its versatile nature, graphene has been incorporated into lithium-ion batteries, lithium-sulphur batteries, supercapacitors and fuel cells, of which there are multiple variations of each available on the market today.
Check out our detailed Graphene Batteries User’s Guide here.
Electron Emission Displays
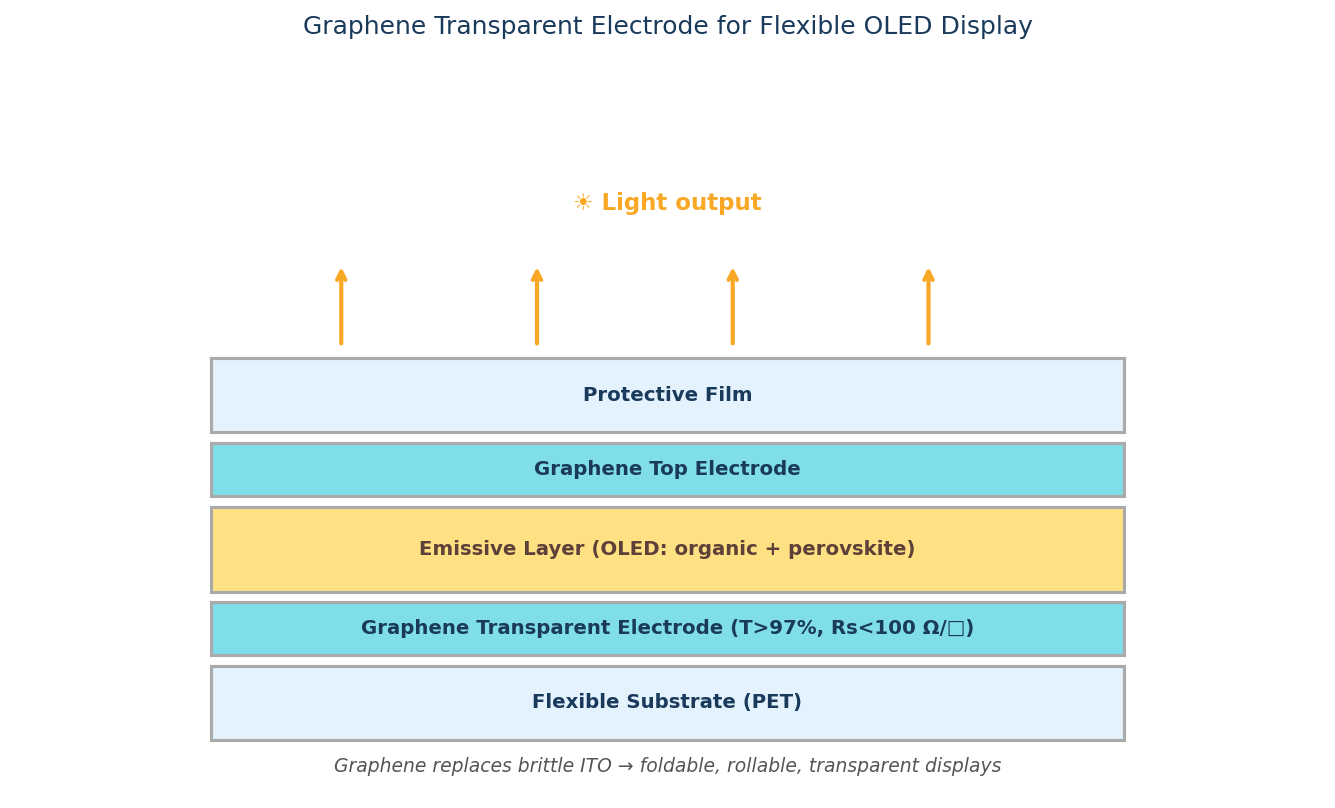
Graphene is an ideal material for use in electron emission displays as it exhibits a high aspect ratio and the dangling bonds at either end of the sheet allow for efficient electron tunneling.
The linear disperisty that the graphene surface provides produces massless Dirac Fermions. When exposed to an electric field, the field emission liberated electrons avoid all back-scattering because their escape velocity is independent to their energy.
Graphene can turn-on an electric field at 0.1 V µm-2, with a field enhancement factor of up to 3700. This can increase up to 4500 in screen printed graphene films.
Structural Composites
Graphene is incorporated into various composites for applications where strength and weight are limiting factors, for example in the aerospace industry.
Graphene is being incorporated into many materials to make the existing material stronger and more lightweight. For the aviation industry, a composite material which is much lighter than steel but will still provide the necessary strength will save a lot of money on fuel consumption, which is why graphene has started to be incorporated into such materials.
Graphene-based structural composites have a huge potential to become a widely used alternative to many materials used today.
Catalyst Supports
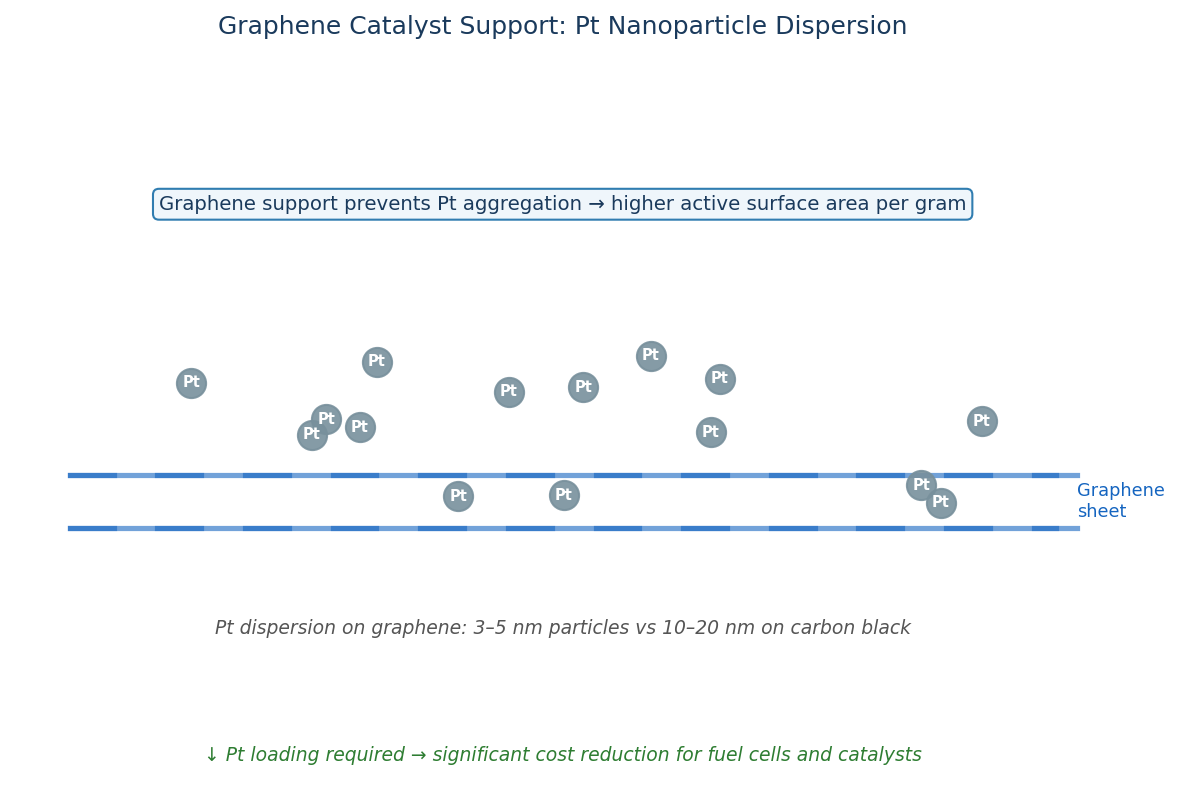
Even though the surface of graphene is planar and uniform, like any other material in existence it is subject to intrinsic defects.
Catalysts in the form of metal ions can sit in these cavities and be supported. In addition to providing mechanical support, the excellent charge carrier ability of graphene assists the charge transfer reactions involving the catalyst.
Graphene is also inert and does not interfere (in a negative way) with the interaction between the catalyst and the substrate materials. Graphene also provides an even dispersion of catalyst particles, so the catalyst-substate reaction is consistent across the whole support.
Polymer Masterbatches
Graphene can be incorporated into polymeric materials to form graphene-polymer composite materials.
As many polymeric materials suffer from strength-related problems, the incorporation of graphene can help to increase the tensile strength of the polymers, increasing the shelf life of the polymeric material in commercial applications.
Incorporating graphene into polymers can also give polymers electrical conductivity properties.
Functional Inks
Graphene can be used in functional inks for electronic, heat resistant and anti-corrosion purposes. By incorporating graphene into ink formulations, the conductivity properties associated with graphene influence the ink, causing it to become conductive.
The inks can then be used to coat electronics. Compared to other conducting inks, graphene is non-toxic, environmentally friendly, cheaper, quick-drying and recyclable. Graphene also has a high thermal stability, making it an ideal for heat resistant ink coating in electronic applications that produce large amounts of heat.
It is also an ink of choice when processing temperatures need to be high, as the graphene won’t break down during the manufacturing process. Graphene also exhibits excellent chemical stability and is inert.
For applications where environmental factors are an issue, graphene inks can provide a stable barrier to protect materials from chemicals and corrosion.
We hope you enjoyed this guide and found it informative. Graphene’s next killer app could be Your’s.
References:
Huang X., Xiaoying Q., Boey F. and Zhang H., Graphene based composites, Chem Soc. Rev., 2012, 41, 666-686
Zhou G., Yin L., Wang D. and Cheng H., A fibrous hybrid of graphene and sulfur nanocrystals for high performance lithium-sulfur batteries, ACS Nano, 2013, 7(6)
Cheng Q., Tang J., Zhang H., Graphene and carbon nanotube composite electrodes for supercapacitors with ultra-high energy density, Phys. Chem. Chem. Phys., 2011, 13, 17615-17624
Peng Z., Xiang C., Yan Z., Natelson D., Graphene Nanoribbon and Nanostructured SnO2 Composite Anodes for Lithium Ion Batteries, ACS Nano, 2013, 7(7)
Haegyeom K., Dong-Hwa S., Sung Wook None K., Kisuk K., Highly reversible Co3O4/graphene hybrid anode for lithium rechargeable batteries, Carbon, 2011, 49(1), 326-332
Bak S., Kim D., Lee H., Graphene quantum dots and their possible energy applications: A review, Current Applied Phyics, 2016, 11, 1192-1201
Liu Y., Dobrinksy A., Yakobson B. I., Graphene edge from armchair to zigzag: The origins of nanotube chirality, Phys. Rev. Lett., 2010, 105, 235502
Begliarbekov M., Sasaki K., Sul O., Yang E., Strauf S., Nano Lett., 2011, 11(11), 4874-4878
Pop E., Varshney V., Roy A., Thermal properties of graphene: Fundamentals and applications, MRS bulletin, 2012, 37, 1273-1281
Lei W., Li C., Cole M., Qu K., Ding S., Zhang Y., Warner J., Zhang X., Wang B., Milne W., A graphene -based large area surface-conduction electron emission display, Carbon, 2013, 56, 255-263
www.cheaptubes.com
www.graphenea.com
Global Graphene Battery Market Worth USD 115M Report (BusinessWire, 2016)
http://s3.amazonaws.com/academia.edu.documents/41175514/Advanced_carbon_aerogels_for_energy_appl20160114-15050-1liyorj.pdf?AWSAccessKeyId=AKIAJ56TQJRTWSMTNPEA&Expires=1480256913&Signature=21XHgBv83B69AOLeWNHLpXxdIWs%3D&response-content-disposition=inline%3B%20filename%3DAdvanced_carbon_aerogels_for_energy_appl.pdf
KAIST Institute for NanoCentury — Graphene Research Highlight
http://www.4spepro.org/pdf/004401/004401.pdf
Pushing Graphene (McEuen Group, Cornell University — JVSTB)
Pop E. — Thermal properties of graphene: Fundamentals and applications (arXiv:1301.6181)
Graphene Sensors Applications (University of Manchester)
New Graphene-Based Inks for Printed Electronics (University of Cambridge)
Source Research-Grade Graphene from Specialists
We supply CVD graphene films, graphene oxide, reduced graphene oxide, and graphene nanoplatelets — every material covered in this guide. Consistent quality, CoA with every order, and technical support from a team with 20 years in nanomaterials.
