TL;DR: Fullerenes are hollow-cage carbon allotropes — C60 (the soccer-ball Buckminsterfullerene, MW 720.64 g/mol, icosahedral symmetry) and C70 (ellipsoidal, MW 840.74 g/mol) are the most widely used. They’re synthesized by arc-evaporating graphite rods in helium (the Kratschmer-Huffman method), then extracted with aromatic solvents and purified by HPLC. Fullerenes and their derivatives (PCBM, PC71BM, fullerenols, endohedral metallofullerenes) are benchmark electron acceptors in organic photovoltaics, n-type semiconductors in OFETs, molecular-scale lubricants, potent radical scavengers, and platforms for biomedical and energy-storage research.
By Mike Foley, Founder, Cheap Tubes Inc. — published April 2026
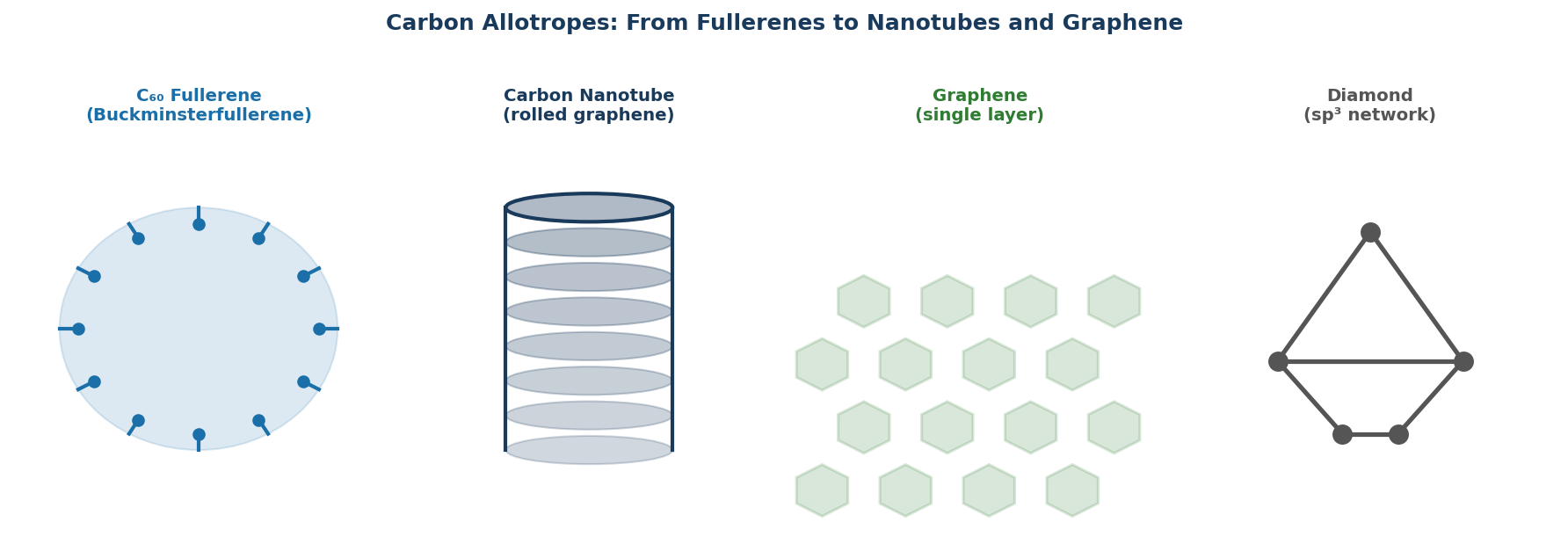
1. What Are Fullerenes?
In 1985, Harold Kroto, Robert Curl, and Richard Smalley published a short paper in Nature describing a new carbon allotrope: 60 carbon atoms arranged in a hollow, soccer-ball-shaped cage. The molecule was named Buckminsterfullerene, after Buckminster Fuller, whose geodesic domes it resembles at the molecular scale. Kroto, Curl, and Smalley shared the 1996 Nobel Prize in Chemistry for the discovery.
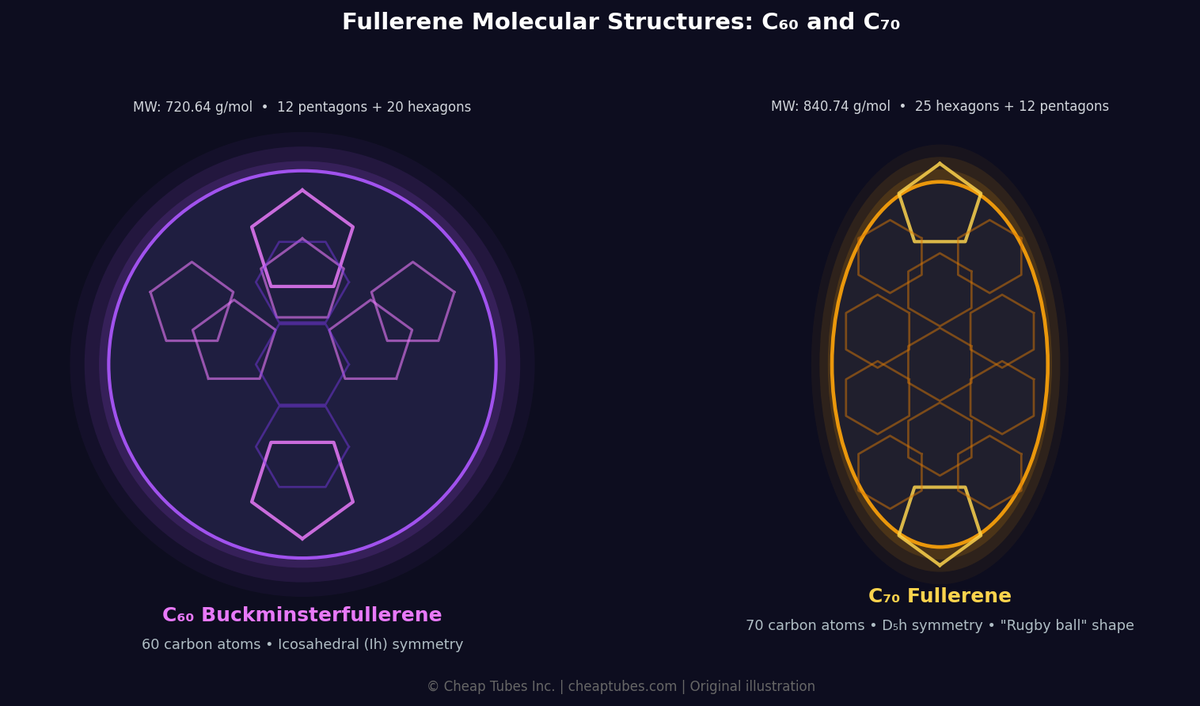
What makes fullerenes distinctive is closed curvature. Graphite is flat sheets of sp² carbon. Diamond is sp³ carbon in a 3D lattice. Fullerenes introduce a third structural paradigm: closed polyhedral cages, where exactly 12 pentagons and a varying number of hexagons wrap the carbon lattice into a finite, hollow molecule. Every fullerene obeys Euler’s formula for polyhedra — C60 has 60 vertices, 90 edges, and 32 faces (12 pentagons + 20 hexagons).
The family includes:
- C60 — Buckminsterfullerene, icosahedral, the canonical fullerene
- C70 — the second-most-abundant natural fullerene, ellipsoidal
- C76, C78, C82, C84 — higher “giant” fullerenes with multiple isomers
- Endohedral fullerenes — fullerene cages with an atom trapped inside (e.g., La@C82, Sc3N@C80)
- Functionalized derivatives — fullerenols, aminofullerenes, PCBM, dendrimer-fullerenes — where surface groups tune solubility and reactivity
Fullerenes are distinct from carbon nanotubes, which extend the cage topology into a tube, and from graphene, which is the infinite 2D sheet. All three share sp² carbon — they’re topological cousins.
2. Structure and Symmetry
C60 structure: Each of the 60 carbon atoms sits at the junction of two hexagons and one pentagon. No pentagon touches another pentagon — this is the Isolated Pentagon Rule (IPR), which governs which fullerene cages are stable. The result is a molecule with full icosahedral (Iₕ) symmetry — the highest symmetry of any molecule. All 60 carbon atoms are chemically equivalent.
C70 structure: C70 has 70 carbon atoms arranged in 12 pentagons and 25 hexagons. The shape is ellipsoidal, elongated along one axis — five additional hexagons form an equatorial belt. Symmetry drops from Iₕ to D5ₕ, and the 70 carbon atoms occupy five distinct chemical environments.
Bonding: C60 has two bond lengths — a shorter “6-6” bond (1.38 Å, between two hexagons, more double-bond character) and a longer “6-5” bond (1.45 Å, between a hexagon and a pentagon, more single-bond character). This bond alternation has consequences for reactivity: the 6-6 bonds are preferential sites for cycloadditions.
Higher Fullerenes — Beyond C60 and C70
Higher fullerenes are lab-relevant but less widely used:
- C76 exists as a single chiral D2 isomer — the first confirmed chiral fullerene. Research interest in molecular chirality and chiral electrochemistry.
- C78 has multiple stable isomers (D3, C2v variants) — used to study isomer-specific optical and electronic properties.
- C82 and C84 — the canonical hosts for endohedral metallofullerenes (e.g., La@C82, Sc3N@C80). Their larger cages accommodate metal atoms and clusters that won’t fit inside C60.
In arc-evaporated crude fullerene extract, C76–C84 typically represent 2–5% of the total fullerene fraction. Separating them from C60/C70 requires more HPLC stages than a C60/C70 separation, with yields of roughly 0.1–1% of starting soot. Pricing runs 10–50× the per-gram cost of C60. Most researchers only source higher fullerenes when the endohedral chemistry or a specific isomer property justifies the cost.
3. Physical and Chemical Properties
C60 properties
| Property | Value |
|---|---|
| Molecular formula | C₆₀ |
| Molecular weight | 720.64 g/mol |
| Cage diameter (outer) | ~0.71 nm |
| Point group | Iₕ (icosahedral) |
| Bond lengths | 1.38 Å (6-6), 1.45 Å (6-5) |
| Density (solid, fcc) | 1.65 g/cm³ |
| Sublimation temperature | ~400 °C (vacuum) |
| Solubility in toluene | ~2.8 mg/mL at 25 °C |
| Solubility in CS₂ | ~7.9 mg/mL at 25 °C |
| Solubility in water | < 10⁻⁹ mg/mL (essentially insoluble) |
| Electron affinity | 2.65 eV |
| Ionization potential | 7.6 eV |
| HOMO-LUMO gap (solid) | 1.9 eV |
| Reversible reduction | up to 6 electrons (C₆₀ᵐ⁻, m = 1–6) |
| Max Li atoms per cage | 12 |
C70 properties (differences from C60)
| Property | C60 | C70 |
|---|---|---|
| Molecular weight | 720.64 g/mol | 840.74 g/mol |
| Point group | Iₕ | D5ₕ |
| Shape | Spherical | Ellipsoidal |
| Solubility in toluene | ~2.8 mg/mL | ~1.4 mg/mL |
| Visible absorption | weaker | broader, stronger |
| OPV active-layer PCE (typical) | baseline | 5–10% higher than C60 |
| Cost | lower | higher (lower natural abundance) |
Reactivity
C60 is a strong electron acceptor — the triply degenerate LUMO can reversibly accept up to 6 electrons in solution, producing the C₆₀⁶⁻ hexaanion. This is the fundamental reason fullerenes are the benchmark electron acceptor in organic photovoltaics: they accept excited-state electrons from donor polymers cleanly.
Three dominant reaction classes:
- Cycloadditions — [4+2] Diels-Alder reactions at 6-6 bonds are the workhorse for making PCBM and related OPV derivatives
- Nucleophilic addition — amines, organolithium reagents, cyanide add to 6-6 bonds
- Radical addition — under photochemistry or with initiators, C60 reacts with dozens of radicals per cage, which is why it’s a potent antioxidant
Fullerenes in solution are photosensitive — UV exposure can drive oligomerization. Store solutions in amber vials, refrigerated, in the dark.
4. Characterization and Purity Verification
Research-grade fullerenes are defined by the purity of the starting material — and purity can only be claimed if it’s measured. Standard characterization methods:
HPLC (high-performance liquid chromatography) — the reference method. Reverse-phase columns with toluene/hexane or pure toluene mobile phase, UV detection at 330 nm (C60) or 390 nm (C70). A research-grade C60 sample shows a dominant C60 peak with ≤ 1% area fraction from contaminants (C70, higher fullerenes, or residual solvent). HPLC is also the primary tool for separating C60 from C70 and from the higher fullerenes in crude extract.
UV-Vis spectroscopy — pure C60 in toluene has a characteristic absorption spectrum with peaks at 329 nm, 409 nm, and a weaker band in the 540–620 nm range. Deviations indicate contamination or oxidation.
¹³C NMR — C60 has all 60 carbons chemically equivalent, so ¹³C NMR shows a single peak at ~143 ppm. C70 has 5 inequivalent carbons, giving 5 peaks in the 130–150 ppm region. Clean check for isomeric purity.
MALDI-MS — matrix-assisted laser desorption/ionization mass spectrometry with minimal matrix interference gives clean C60⁺ (m/z 720) or C70⁺ (m/z 840) peaks. Higher fullerene contaminants appear at expected m/z values (C76 at 912, C78 at 936, C84 at 1008).
IR spectroscopy — C60 has exactly 4 IR-active modes (T1ᵤ symmetry): 1429, 1183, 577, 527 cm⁻¹. An IR spectrum with only these four lines is strong evidence of pure, non-functionalized C60. Extra bands usually indicate oxidation (C-O stretches near 1100 cm⁻¹) or functionalization.
When Cheap Tubes ships research-grade C60 or C70, the Technical Data Sheet includes HPLC purity data and the lot’s characterization fingerprint so you can verify consistency across batches.
5. Synthesis: The Kratschmer-Huffman Method
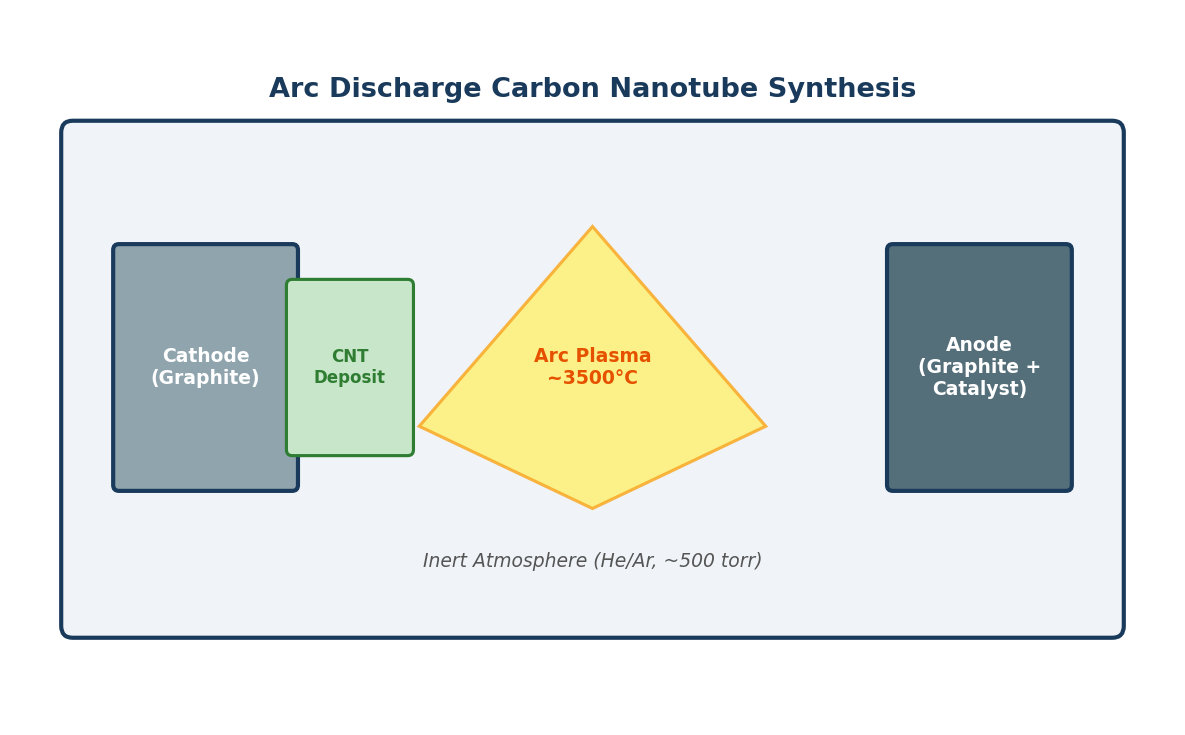
Fullerenes are formed at gram-scale abundance in soot produced by vaporizing graphite in an inert atmosphere. The practical synthesis most suppliers (including Cheap Tubes) source from is the Kratschmer-Huffman arc-evaporation method, published in 1990 and essentially unchanged:
- Arc: Two graphite electrodes sit in a chamber filled with helium at ~100 Torr. A high DC current (~100 A) strikes an arc, vaporizing the graphite.
- Soot formation: Carbon atoms and small clusters condense in the inert helium flow, self-assembling into fullerenes and fullerene-like structures. The resulting soot contains roughly 10–15% fullerenes by mass, of which ~85% is C60, ~10% is C70, and a few percent is C76/C78/C84.
- Extraction: The soot is Soxhlet-extracted with toluene or ortho-dichlorobenzene. Fullerenes dissolve; most of the amorphous carbon doesn’t.
- Purification: Extracted material is fractionated on a column — most commonly reverse-phase HPLC with toluene/hexane mobile phase — to separate C60 from C70 and higher fullerenes.
- Drying and verification: Purified fullerene is crystallized from toluene, dried under vacuum, and assayed by HPLC (and optionally NMR, MS, or IR) for purity — ≥ 99% for research grade, ≥ 99.9% for highest-grade applications.
Alternative methods exist — combustion synthesis (Frontier Carbon’s Nano-C process), laser vaporization, CVD — but arc evaporation remains the commercial default for general C60 and C70.
6. Applications Across Research and Industry
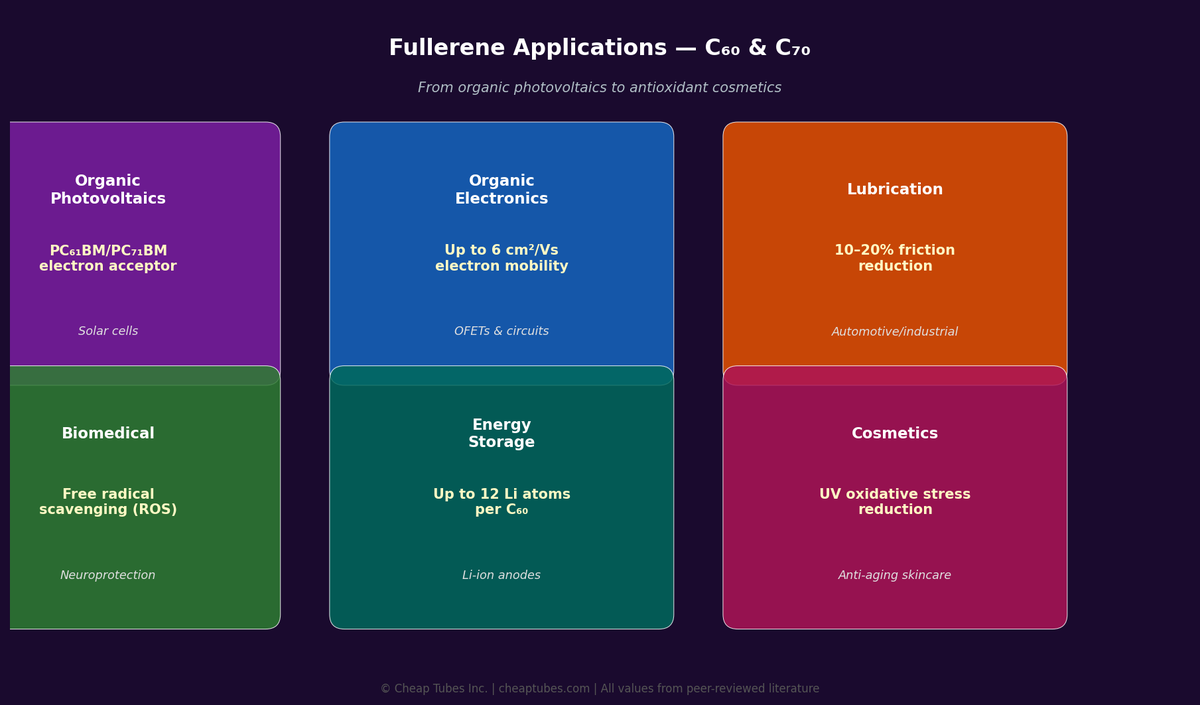
6.1 Organic Photovoltaics (OPV)
Fullerenes and their derivatives are the benchmark electron acceptor in organic solar cells. The two workhorses:
- PCBM ([6,6]-phenyl-C₆₁-butyric acid methyl ester) — the C60 derivative that enabled efficient bulk heterojunction OPV. Toluene- and chlorobenzene-soluble, miscible with donor polymers like P3HT and PTB7.
- PC71BM — the C70 analog. Stronger visible absorption boosts current generation, giving 5–10% higher PCE than PC61BM in matched architectures.
Current OPV records using fullerene acceptors reach 11–13% PCE in small-area cells.
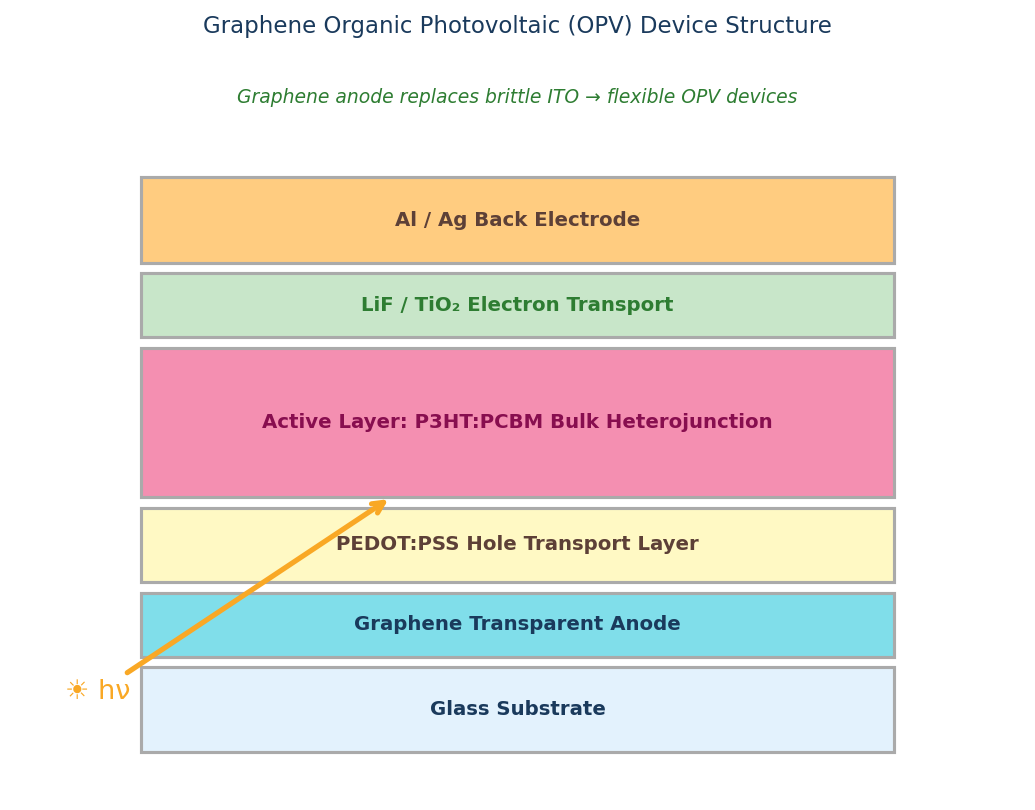
Fullerenes vs Non-Fullerene Acceptors (NFAs)
Since about 2018, non-fullerene acceptors — particularly Y6 and its derivatives (L8-BO, BTP-eC9) — have surpassed fullerene-based OPVs in peak power conversion efficiency. Current single-junction NFA records exceed 19% PCE, where fullerene-based cells top out around 11–13%.
So why are fullerenes still widely used?
- Reference and benchmarking — PCBM/PC71BM are the electron acceptors against which new donor polymers are characterized. Virtually every OPV paper reports both NFA and fullerene performance for the same donor.
- Morphology control — fullerene acceptors crystallize and pack more predictably than Y6-family molecules, enabling reliable bulk-heterojunction morphologies for device physics studies.
- Tandem architectures — fullerene subcells are still common in multi-junction devices.
- Long-term stability research — fullerene-based OPVs have a longer documented stability literature.
- Low-cost exploratory work — C60 and PCBM are cheaper and synthetically simpler than Y6-family NFAs.
For researchers: fullerenes are the known-good reference system; NFAs are the cutting edge. Most modern OPV programs use both.
6.2 Organic Field-Effect Transistors (OFETs)
Thermally evaporated C60 films are among the highest-mobility n-type organic semiconductors ever measured. Electron mobilities up to 6 cm²/V·s have been reported in purified, single-crystalline C60 transistors. Used in:
- Flexible CMOS logic (pairing with p-type pentacene)
- Chemical and biological sensors
- Light-emitting transistors
- Organic thermoelectric devices
6.3 Lubrication and Tribology
C60’s near-spherical geometry and low intermolecular friction make it a molecular ball bearing. Fullerene lubricant additives reduce friction coefficients by 10–30% in metal-on-metal contacts, particularly under high-pressure or high-vacuum conditions where conventional organic lubricants decompose. C60 is used in ultra-high-vacuum precision bearings and as a solid-lubricant additive in spacecraft mechanisms.
6.4 Radical Scavenging and Antioxidants
C60 is arguably the most potent radical scavenger known in condensed matter. Its 3D π-system reacts with up to 34 methyl radicals per molecule and efficiently traps oxygen-centered and carbon-centered radicals. Peer-reviewed studies show C60 in squalane outperforms vitamin C and vitamin E in UV-oxidative stress assays on skin cells. This has driven its use in premium anti-aging formulations.
6.5 Biomedical Research
Fullerenes — particularly water-soluble derivatives like fullerenols (poly-hydroxylated C60) and aminofullerenes — are active research subjects for:
- Antiviral activity: HIV protease inhibition; more recently, SARS-CoV-2 main protease binding studies
- Photodynamic therapy (PDT): Fullerene derivatives generate singlet oxygen under visible light, useful for targeted cancer cell killing
- Antibacterial coatings: Fullerene-functionalized surfaces in medical devices
- Neuroprotection: Radical-scavenging activity studied in Parkinson’s and Alzheimer’s cell and animal models
Regulatory status: fullerene derivatives are not approved drugs. They’re research-only materials.
6.6 Energy Storage
C60 can reversibly accommodate up to 12 lithium atoms per molecule (Li₁₂C₆₀) — a structural feature that drew early attention for Li-ion anodes. In practice, graphite and silicon dominate commercial Li-ion cells on volumetric performance. Fullerenes remain most relevant in energy-storage research as model systems for electron-transfer kinetics, molecular anodes in hybrid electrodes, and as additives that modify solid-electrolyte interphase formation.
Alkali-metal-doped fullerenes exhibit superconductivity: K₃C₆₀ has a critical temperature Tc = 18 K, and Cs₃C₆₀ reaches Tc = 40 K under pressure — remarkable for a molecular solid.
Endohedral metallofullerenes — a metal atom or cluster trapped inside the cage — are under active study as qubits for molecular quantum information: the encapsulating cage magnetically isolates the internal spin from environmental noise.
7. Derivatives and Functionalization Chemistry
Pure C60 and C70 are useful, but functionalization dramatically expands their application space by modifying solubility, reactivity, and electronic structure.
The dominant reaction sites are the 6-6 bonds — the shorter, more double-bond-like bonds between adjacent hexagons. Two functionalization methods account for most published fullerene chemistry:
Bingel-Hirsch cyclopropanation — a malonate ester is deprotonated to form a carbanion, which attacks a 6-6 bond and cyclopropanates the cage. Adds a three-membered ring fused to a 6-6 edge. Reliable, clean, high-yielding. Used to make PCBM.
Prato 1,3-dipolar cycloaddition — a carbonyl compound (often an aldehyde) and an amino acid (typically sarcosine) condense in situ to form an azomethine ylide, which undergoes [3+2] cycloaddition across a 6-6 bond, yielding a pyrrolidine-fused fullerene. Prato reactions tolerate broader functional groups than Bingel and are the standard route to fullerene derivatives for biomedical use.
Regioselectivity — monofunctionalization (one addition per cage) is usually straightforward, but multifunctionalization (2–6 additions per cage) raises selectivity questions. The “equatorial” and “polar” addition patterns of a bis-functionalized C60 have distinct electronic properties.
Common derivatives:
- PCBM / PC71BM — methano-fullerenes made by Bingel chemistry, workhorse OPV acceptors (see section 6.1)
- Fullerenols (C₆₀(OH)ₙ, n = 20–40) — water-soluble, biocompatible, used in biomedical research
- Aminofullerenes — C60-NH₂ and amino-pyrrolidine derivatives, used in drug delivery and antiviral research
- Endohedral metallofullerenes — M@C2n where M is a lanthanide or early transition metal, plus tri-metallic nitride endohedrals (Sc3N@C80)
- Fullerene dendrimers — used as scaffolds for controlled multivalent display of bioactive groups
8. Choosing the Right Purity Grade
Not every application needs 99.9% purity. In 21 years of providing research-grade fullerenes, we’ve seen some customers pay for higher grades than they actually need, and others push 98% material through experiments where 99.9% would have achieved their desired results. We are always available to help guide our clients with fullerene selection. General guidance:
- 95% purity — suitable for bulk exploratory work, composite additive research, and antioxidant formulations where trace C70 in C60 is acceptable. Cost-effective for gram-scale chemistry.
- 98% purity — the workhorse grade for most OPV device fabrication, friction/lubrication studies, and photochemistry. Most published OPV device papers report C60 ≥ 98%.
- 99% purity — required for quantitative photophysics (transient absorption, precise triplet yields), detailed electron-transfer kinetics, and device reliability studies where C70 trace contamination skews performance metrics.
- 99.9% purity — demanded for OFET fabrication where carrier mobility is the headline number, endohedral metallofullerene synthesis (each chemistry step amplifies trace contaminants), NMR reference standards, and any work where sub-percent concentration effects matter.
Practical rule: pick the grade where your bottleneck step starts dominating signal-to-noise. Higher purity doesn’t help if your downstream chemistry or device fabrication reintroduces contaminants.
9. Solubility and Handling
Practical notes for working with fullerenes:
- Storage: Air-stable as dry powder. Solutions are UV-sensitive — amber glass, refrigerated, dark.
- Solvent choice: Toluene, ortho-dichlorobenzene, and CS₂ are the workhorse solvents for native C60/C70. For functionalized derivatives, polarity tunes solubility: fullerenols are water-soluble, PCBM is moderately polar and dissolves in chlorobenzene + toluene.
- Purity verification: HPLC is the reference — a research-grade C60 sample should show a dominant single peak with ≤ 1% area fraction from contaminants. Request the lot’s HPLC chromatogram from your supplier if you don’t receive one.
- Safety: Acute toxicity of pure C60 is very low (LD50 > 2 g/kg in rodent models). Functionalized derivatives have variable profiles — consult the specific product’s SDS. Standard nanomaterial handling applies: dust containment, nitrile gloves, lab coat, appropriate PPE.
10. Frequently Asked Questions
What is the difference between a fullerene and a carbon nanotube?
Both are sp² carbon cage structures, but fullerenes are finite, closed cages (C60, C70, etc.), while carbon nanotubes extend the cage topology into a hollow tube — in principle infinitely long. A single-walled nanotube is effectively a “rolled-up” graphene sheet; a fullerene is a spherical-ish “rolled-up and closed” sheet.
What is PCBM, and why is it used instead of raw C60?
PCBM ([6,6]-phenyl-C₆₁-butyric acid methyl ester) is a C60 derivative with a methanofullerene sidegroup that dramatically increases solubility in common organic solvents (toluene, chlorobenzene). Raw C60 is poorly soluble in the solvents used for solution-processed OPV active layers, so PCBM is the practical workhorse. It’s made by Bingel cyclopropanation of C60 with a methyl ester-substituted malonate.
Can I use fullerenes for batteries?
Research-only — C60 reversibly accepts up to 12 lithium atoms per molecule, but volumetric energy density is lower than graphite or silicon. Fullerenes are most relevant in battery research as model systems for studying electron-transfer kinetics, as additives in hybrid electrode formulations, or as templates for studying solid-electrolyte interphase (SEI) chemistry.
Are fullerenes safe to handle?
Pure C60 has very low acute toxicity in rodent studies (LD50 > 2 g/kg). Functionalized derivatives vary — always consult the specific product’s SDS. Standard nanomaterial handling (dust containment, nitrile gloves, lab coat, appropriate PPE) applies. Our Carbon Nanotube Safety Data Sheet page has general nanomaterial safety context.
How is fullerene purity measured?
HPLC is the reference method — reverse-phase with toluene/hexane mobile phase, UV detection at 330 nm (C60) or 390 nm (C70). Supplementary techniques include ¹³C NMR (single peak for C60 at ~143 ppm), UV-Vis spectroscopy, MALDI mass spectrometry (clean C60⁺ at m/z 720), and IR spectroscopy (4 modes for pure C60).
Why are endohedral metallofullerenes interesting?
A metal atom (or small cluster) trapped inside the carbon cage is shielded from the chemical environment by the cage wall. This makes endohedrals promising as molecular qubits — the internal spin sees a much quieter magnetic environment than it would in a free ion. They’re also of interest as MRI contrast agents and for studying atom-cage charge transfer.
Where can I buy research-grade C60 and C70?
Cheap Tubes supplies research-grade C60 (99%+), C70 (99%+), and ultra-high purity C60 (99.9%) with TDS and SDS included on every order. See the fullerenes product catalog for current options and pricing.
Related Reading
- Solar Applications of Graphene — OPV and thin-film photovoltaic research with sp² carbon materials
- Graphene Batteries: An Insider’s Guide — how conjugated carbon materials are being engineered for energy storage
- Carbon Nanotube Composites: Types, Properties & Uses — related sp² carbon architectures in composite formulations
- Nanotechnology Glossary — terminology reference
References
Canonical literature on fullerenes:
- Kroto, H. W.; Heath, J. R.; O’Brien, S. C.; Curl, R. F.; Smalley, R. E. Nature 1985, 318, 162. (original C60 discovery)
- Krätschmer, W.; Lamb, L. D.; Fostiropoulos, K.; Huffman, D. R. Nature 1990, 347, 354. (arc-evaporation synthesis)
- Hirsch, A.; Brettreich, M. Fullerenes: Chemistry and Reactions. Wiley-VCH, 2005. (textbook reference for chemistry)
- Yu, G.; Gao, J.; Hummelen, J. C.; Wudl, F.; Heeger, A. J. Science 1995, 270, 1789. (first OPV bulk heterojunction)
- Bingel, C. Chem. Ber. 1993, 126, 1957. (Bingel cyclopropanation)
- Maggini, M.; Scorrano, G.; Prato, M. J. Am. Chem. Soc. 1993, 115, 9798. (Prato cycloaddition)
- Yuan, J.; Zhang, Y.; Zhou, L.; et al. Joule 2019, 3, 1140. (Y6 NFA reference)
Author
Mike Foley founded Cheap Tubes Inc. in Townshend, Vermont in 2005 to supply researchers and R&D engineers with spec-guaranteed carbon nanotubes, graphene, and advanced nanomaterials. Over 21 years, Cheap Tubes has shipped research-grade materials to more than 10,000 customers, including research groups at MIT, NASA, Rice, Harvard, 3M, and the US Army. Mike has personally QC’d tens of thousands of fullerene orders and consults with OPV, lubrication, and biomedical labs on material selection.
Contact: Request a quote | sales@cheaptubes.com
