Functionalized Carbon Nanotubes
Quick Summary: Functionalized Carbon Nanotubes
- What they are — pristine SWCNT or MWCNT with reactive surface groups (COOH, OH, NH₂) added by chemical treatment
- Why they matter — dramatically improved dispersion in water and polar solvents, plus reactive handles for conjugation and matrix bonding
- Compared to pristine CNTs — better dispersibility, easier to formulate, slightly lower aspect ratio after acid oxidation
- Key applications — biomedical scaffolds, drug delivery, polymer composites, conductive inks, sensors, dispersion-critical formulations
- Available chemistries — COOH (carboxyl), OH (hydroxyl), NH₂ (amine)
- Choosing the right one — see the SWCNT Buying Guide for functionalization selection by application
Functionalized Carbon Nanotubes
Pristine CNTs won’t bond to your matrix. They aggregate in water. They have no reactive handles for your conjugation chemistry. Functionalization solves all of this — but only when the surface chemistry is matched to your specific application and matrix.
Cheap Tubes has supplied COOH, OH, NH₂, F, and nitrogen-functionalized SWCNTs and MWCNTs to research labs and development teams since 2005 — acid-functionalized and plasma-functionalized grades, with representative characterization data for each. Used in drug delivery, composite reinforcement, biosensor development, and specialty polymer applications. If our material doesn’t meet published specifications, we’ll replace it or refund your order. Ships from Vermont.
In stock. Contact us for custom functionalization density, surface group ratios, or volume pricing.
Used in published research: Pharmaceutics 2021 · Biology 2021
Showing 1–12 of 51 results
-

COOH Functionalized Graphitized Multi Walled Carbon Nanotubes 10-20nm
$6.00 - $35.00 / per gram Select options -

COOH Functionalized Graphitized Multi Walled Carbon Nanotubes 20-30nm
$3.50 - $30.00 / per gram Select options -

COOH Functionalized Graphitized Multi Walled Carbon Nanotubes 30-50nm
$3.49 - $25.00 / per gram Select options -

COOH Functionalized Graphitized Multi Walled Carbon Nanotubes 50nm
$2.80 - $25.00 / per gram Select options -
COOH Functionalized Graphitized Multi Walled Carbon Nanotubes 8-15nm
$4.49 - $45.00 / per gram Select options -

COOH Functionalized Multi Walled Carbon Nanotubes 10-20nm
$1.35 - $18.00 / per gram Select options -

COOH Functionalized Multi Walled Carbon Nanotubes 20-30nm
$0.99 - $17.00 / per gram Select options -
COOH Functionalized Multi Walled Carbon Nanotubes 20nm
$2.50 - $30.00 / per gram Select options -

COOH Functionalized Multi Walled Carbon Nanotubes 30-50nm
$0.60 - $15.00 / per gram Select options -

COOH Functionalized Multi Walled Carbon Nanotubes 50nm
$0.60 - $15.00 / per gram Select options -
COOH Functionalized Multi Walled Carbon Nanotubes 8-15nm
$2.42 - $18.00 / per gram Select options -

COOH Functionalized Multi Walled Carbon Nanotubes 8nm
$2.49 - $25.00 / per gram Select options
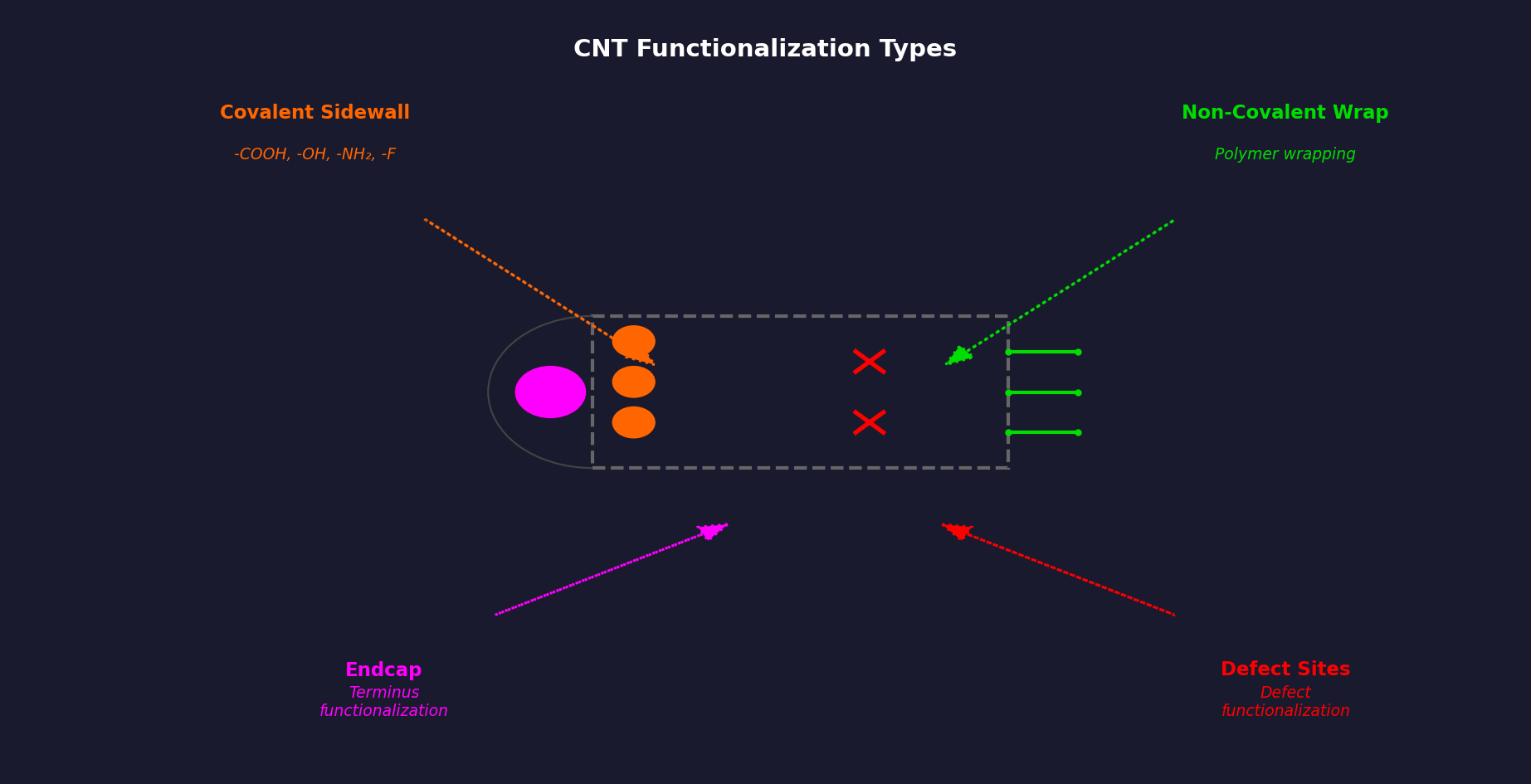
What Is CNT Functionalization?
Carbon nanotube functionalization is the controlled attachment of chemical groups to nanotube surfaces, either covalently (forming direct C–X bonds to the sp² carbon lattice) or non-covalently (through π–π stacking, wrapping, or physisorption). The purpose is to convert pristine CNTs — chemically inert, hydrophobic, and strongly bundled — into CNTs that can be dispersed in specific solvents or matrices, bond to target surfaces, or participate in chemical reactions. The type and density of surface groups determines which applications become accessible.
Types of Functionalized CNTs
| Functional Group | Symbol | Key Properties | Primary Applications |
|---|---|---|---|
| Carboxylic acid | -COOH | Disperses in water and polar solvents; reactive toward amines and alcohols | Polymer composites (epoxy, PA, PC), biomedical, covalent coupling |
| Hydroxyl | -OH | Moderate polarity; reacts with isocyanates, silanes, epoxides | Polyurethane composites, surface coatings, silane coupling |
| Amine | -NH₂ | Basic, highly water-dispersible; reacts with COOH, epoxides, aldehydes | Drug delivery, biosensors, crosslinked epoxy composites |
| Fluorine | -F | Highly reactive toward nucleophiles; enables further functionalization | Covalent attachment chemistry, fluoropolymer composites |
| Nitrogen-doped | -N, N₂ | Electron donor; improves catalytic activity and electrochemical behavior | Electrocatalysis (ORR), supercapacitors, fuel cells |
Functionalization Methods
Acid Functionalization
The most widely used method treats CNTs with concentrated nitric acid, sulfuric acid, or mixtures of both at elevated temperature. This oxidizes the tube surfaces and ends, introducing carboxylic acid (-COOH) and hydroxyl (-OH) groups primarily at defect sites and tube ends (which are more reactive than intact sidewalls). Acid functionalization also removes residual catalyst metals from CVD synthesis, reducing metal content to <1 wt%. The drawback is that it introduces additional lattice defects and shortens the tubes, which reduces aspect ratio and can lower mechanical properties in composites.
Plasma Functionalization
Plasma treatment exposes CNTs to reactive gas plasmas (oxygen, nitrogen, ammonia, or fluorine plasmas) that attach functional groups to the surface with minimal internal lattice damage. Plasma functionalization is gentler than acid treatment — it preserves tube length and introduces fewer structural defects — while still providing effective surface group attachment and catalyst removal. It is the preferred method when preserving mechanical and electrical properties is important alongside the functionalization.
Where Functional Groups Locate on CNTs
The sp² graphene lattice of CNT sidewalls is relatively chemically inert. Functional groups attach preferentially at higher-energy sites: end caps (which have 5-membered rings), pre-existing lattice defects, and sites of incomplete hexagon closure. This means the density of functional groups is higher at tube ends and defect sites than on pristine sidewalls. The degree of functionalization is typically quantified by the COOH or other group content as a weight percentage, measured by thermogravimetric analysis (TGA).
Benefits of Functionalization for Composite Applications
In polymer matrix composites, the limiting factor for CNT performance is typically the quality of the CNT–matrix interface. Pristine CNTs bond to most polymer matrices only through weak van der Waals forces, producing poor stress transfer and pull-out at low strains. COOH-functionalized CNTs can form covalent ester or amide bonds with epoxy, polyamide, and polyurethane matrices — dramatically improving interfacial shear strength. This translates to 30–60% higher composite tensile modulus and 20–40% higher strength compared to pristine CNT composites at equivalent loading.
For dispersion in aqueous biological or analytical applications, -COOH and -NH₂ groups provide colloidal stability through electrostatic repulsion (high negative or positive zeta potential) without requiring surfactants that would need to be removed in downstream processing. This makes functionalized CNTs the preferred choice for drug delivery systems, biosensors, and diagnostic conjugation chemistry.
Selecting the Right Functionalized CNT Grade
Cheap Tubes offers COOH, OH, NH₂, F, and nitrogen-functionalized grades for both single-walled (SW/DWNT) and multi-walled CNTs, across diameter ranges from 8 nm to >50 nm. Graphitized functionalized grades (COOH on graphitized MWCNTs) provide higher conductivity alongside functional surface groups. Key selection parameters are the degree of functionalization required (light functionalization preserves electrical conductivity; heavy functionalization improves dispersibility at the cost of some conductivity), tube diameter (affects aspect ratio, percolation threshold, and surface-to-volume ratio), and the target matrix chemistry.
For custom functionalization density, specific surface group ratios, or volume pricing, contact our technical team. SDS documentation is available from our SDS page.
Frequently Asked Questions
Why functionalize carbon nanotubes?
What is the difference between -COOH, -OH, and -NH2 functionalization?
Does functionalization affect electrical conductivity?
How is functionalization quantified?
Can I conjugate biomolecules to functionalized CNTs?
Showing 1–12 of 51 results
