By Mike Foley, Founder, Cheap Tubes Inc. & CTI Materials LLC.
Part of the Graphene & CNT Battery Applications hub.
TL;DR
Graphene nanoplatelets, reduced graphene oxide, and MXene (Ti3C2Tx) serve as high-surface-area electrodes in electrochemical double-layer capacitors (EDLCs) and pseudocapacitors. Graphene provides up to 2,630 m²/g theoretical surface area; practical electrodes achieve 200–700 m²/g. MXene contributes both double-layer capacitance and surface-redox pseudocapacitance, achieving volumetric capacitances above 1,500 F/cm³ in optimized films — among the highest reported for any electrode material. This page covers EDLC vs pseudocapacitor selection, material-grade choice, and procurement considerations for graphene and MXene supercapacitor electrodes.
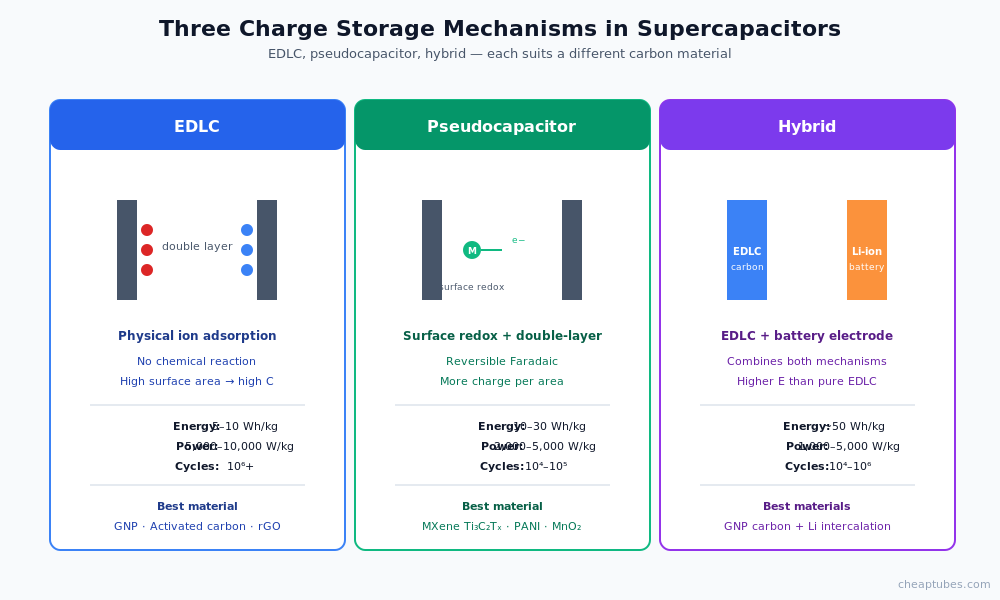
EDLC vs pseudocapacitor vs hybrid
Supercapacitors store charge by three distinct mechanisms, and the right carbon material depends on which mechanism dominates:
Electrochemical double-layer capacitors (EDLCs). Charge is stored physically at the electrode-electrolyte interface — no chemical reaction, just ion adsorption on a high-surface-area substrate. Energy density is modest (5–10 Wh/kg) but power density is very high (5,000–10,000 W/kg) and cycle life is essentially unlimited (106+ cycles). EDLCs use porous carbon, activated carbon, graphene, or rGO as the electrode material.
Pseudocapacitors. Charge is stored via fast, reversible surface redox reactions in addition to double-layer adsorption. Energy density is higher than EDLCs (10–30 Wh/kg) at moderate cost in cycle life (104–105 cycles). Common pseudocapacitive materials: MnO2, RuO2, conductive polymers, and (notably) MXene Ti3C2Tx.
Hybrid supercapacitors. Combine an EDLC electrode (carbon, high power) with a battery-like electrode (lithium intercalation material, high energy). Energy density approaches 50 Wh/kg with power density retained at 1,000–5,000 W/kg. Hybrid devices increasingly dominate commercial supercapacitor product launches in 2024–2026.
The trade-offs determine material choice. For pure EDLC: maximize surface area and conductivity. For pseudocapacitor: choose a material with reversible surface redox at the operating voltage. For hybrid: carbon electrode is typically graphene-based; battery electrode follows lithium-ion design.
Why surface area and conductivity = supercap performance
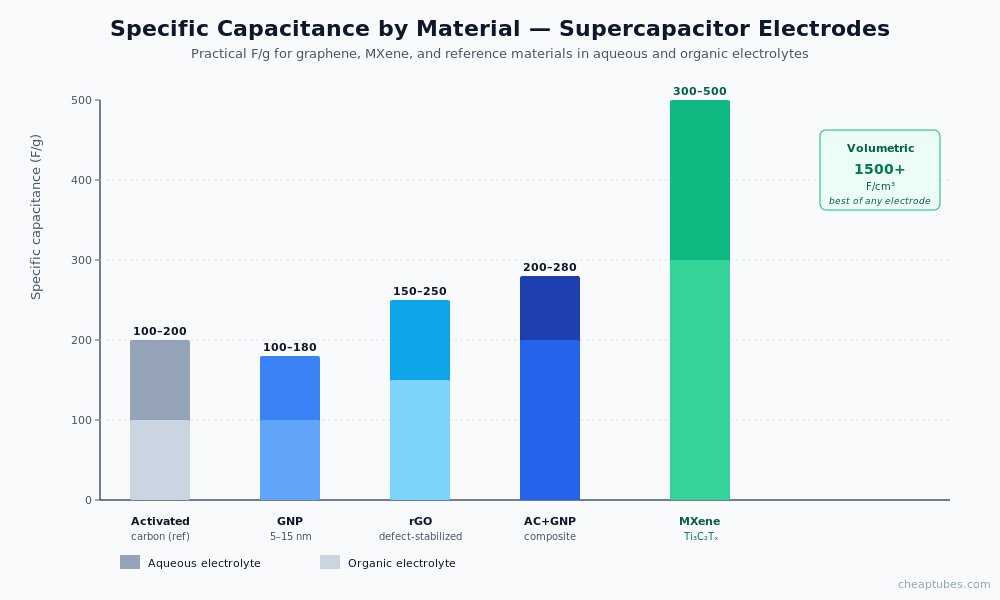
Specific capacitance (F/g) at the EDLC electrode is proportional to electrochemically accessible surface area times the dielectric capacitance of the electrolyte double layer. Practical EDLC carbons deliver 100–250 F/g in aqueous electrolyte and 80–200 F/g in organic electrolyte. Graphene and rGO can exceed 300 F/g in optimized configurations.
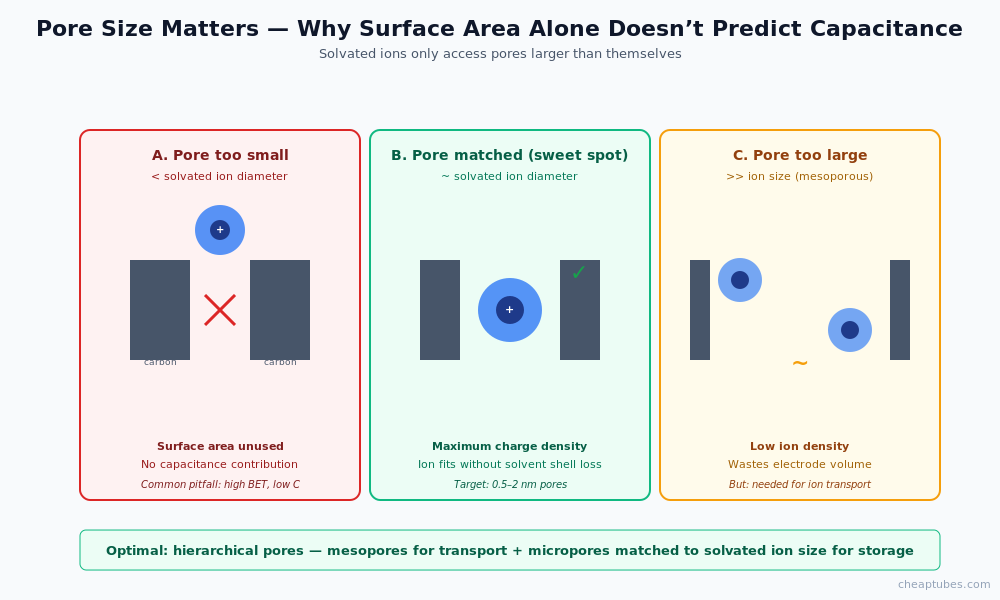
Two surface-area caveats matter:
- Pore size matters more than total surface area. Pores smaller than the solvated ion (1–2 nm for organic electrolytes, 0.5–1 nm for aqueous) contribute little to capacitance because ions cannot access them. Optimal carbon electrodes have a hierarchical pore structure: meso-pores for ion transport plus narrow micro-pores tuned to the solvated ion size for high-density adsorption.
- Conductivity sets the rate ceiling. A high-surface-area carbon with poor conductivity delivers excellent low-rate capacitance but cannot maintain it at high rates. Graphene and MXene have intrinsic in-plane conductivities orders of magnitude higher than activated carbon, which is the reason they dominate high-power supercapacitor research.
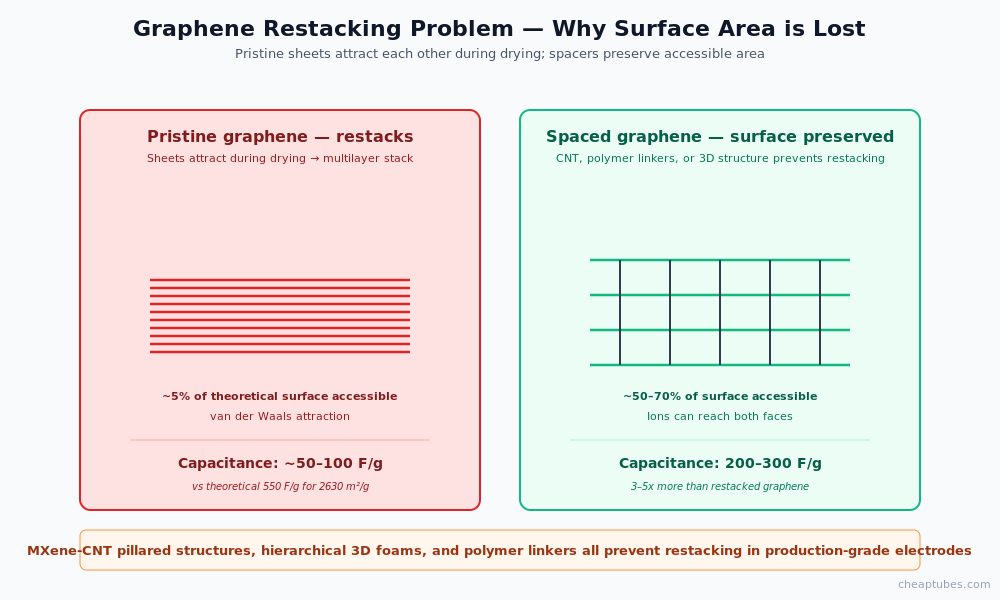
For graphene specifically, the practical challenge is that graphene sheets restack into multilayer assemblies during electrode fabrication — losing most of the theoretical surface area. Successful graphene supercapacitor electrodes use spacers (curved sheets, nanotube intercalation, polymer linkers) to prevent restacking and maintain accessible surface area.
Material-by-material breakdown
Graphene nanoplatelets (GNP) and few-layer graphene. Conductive backbone for EDLC electrodes. Practical specific capacitances 120–200 F/g in aqueous electrolyte at moderate rates. Lower capacitance than activated carbon at low rates, but far higher rate capability and far better cycle stability. Common pairing: GNP + activated carbon hybrid composite, which combines GNP’s conductivity with activated carbon’s surface area. Available from Cheap Tubes’ GNP catalog.
Graphene oxide (GO). Used primarily as a precursor for reduced graphene oxide electrodes. GO itself is electrically insulating because of the oxygen functional groups; it must be reduced to rGO before use as a supercap electrode. GO’s solubility in water and aqueous binders makes it the easiest graphene-family material to process at scale. Available via Cheap Tubes’ GO catalog.
Reduced graphene oxide (rGO). Conductivity restored, defect density tunable via reduction conditions. Practical capacitances 150–250 F/g. rGO defects are sometimes beneficial — they introduce pseudocapacitive contributions from residual oxygen groups, raising total capacitance. rGO is the most common graphene-family electrode in published supercapacitor work.
MXene Ti3C2Tx. The standout 2D material for pseudocapacitors. Layered structure with intercalated water and surface-terminating groups (-O, -OH, -F denoted Tx) that participate in reversible surface redox. Volumetric capacitances exceed 1,500 F/cm³ in dense films — among the highest reported for any electrode material. Areal capacitances of 1–2 F/cm² in thick coatings. Limitations: oxidative degradation in ambient air over long storage; performance dependent on synthesis-induced surface chemistry. Available via Cheap Tubes’ MXene category.
Composite electrodes. Most published high-performance supercap electrodes combine two or more carbon morphologies — for example, GNP backbones interleaved with activated carbon spacers, or MXene + CNT pillared structures that prevent MXene restacking. Hybrid electrodes are the current state-of-the-art for commercial-grade performance.
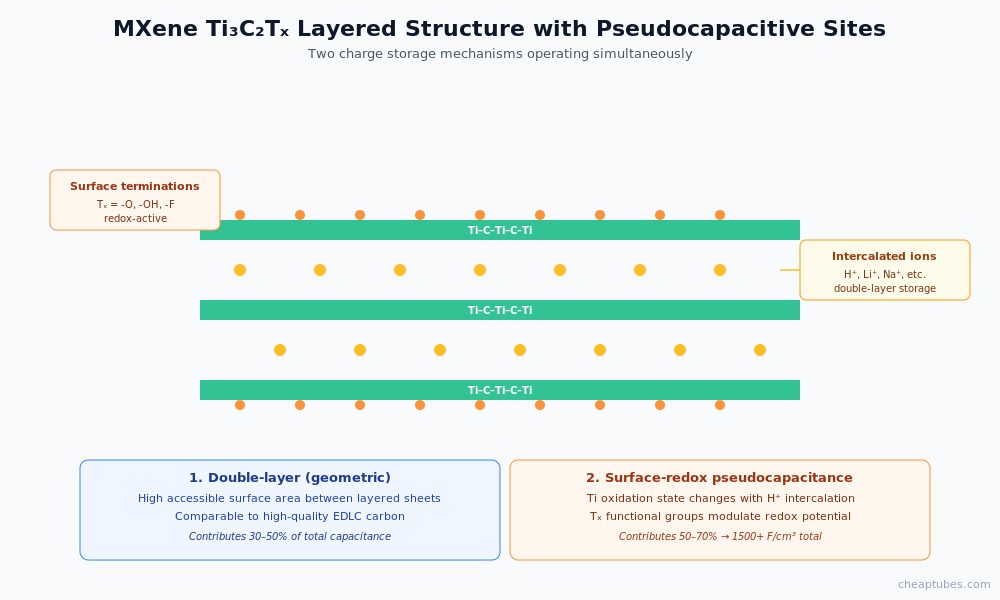
Pseudocapacitive contributions of MXene
MXene’s high volumetric capacitance comes from two simultaneous mechanisms:
- Double-layer capacitance from the high accessible surface area between layered Ti3C2Tx sheets — comparable to a high-quality EDLC carbon.
- Surface-redox pseudocapacitance from oxidation-state changes of surface Ti atoms in the presence of protons or other cations. The functional groups (-O, -OH, -F) on the MXene surface modulate the redox potential and the available redox capacity. This is the contribution that distinguishes MXene from a pure-EDLC graphene electrode.
The combined effect is volumetric capacitance several times that of activated carbon or pristine graphene, with rate capability that scales with the in-plane MXene conductivity. The technology trade-offs: MXene cost is higher than graphene-family materials, and ambient-air stability is more limited (storage and processing require inert or controlled atmospheres for the highest-quality grades).
For supercapacitor R&D and commercial development teams evaluating MXene, the practical specifications to check are: Ti3C2Tx layer count (mono vs few-layer), surface termination distribution (-O, -OH, -F ratio), and pre-intercalation state (delaminated vs multilayer).
Material specs for supercap electrodes
| Material | Practical capacitance (F/g) | Best application | Notes |
|---|---|---|---|
| GNP (5–25 µm lateral, 5–15 nm) | 100–180 | EDLC conductive backbone | Restacking is the main loss mechanism |
| rGO (0.5–5 µm lateral) | 150–250 | EDLC + mild pseudocap | Defect-stabilized, easy to process |
| GO | (precursor only) | Reduce to rGO in situ | Water-soluble, easy slurry processing |
| MXene Ti3C2Tx | 300–500 (mass), 1,500 F/cm³ (vol) | Pseudocapacitor, hybrid | Highest volumetric capacitance; air-stability caveat |
| Activated carbon + GNP composite | 200–280 | Commercial EDLC | Best cost-performance for production |
Loading in the final electrode is typically 70–95 wt% active carbon material, 5–15 wt% binder (PTFE or PVDF), 0–10 wt% conductive additive (carbon black or CNT). For composite electrodes, mass ratios of the carbon components are typically optimized empirically.
For pilot or production-scale electrode coating, dispersion uniformity again drives yield. Surfactant-stabilized Flexiphene formulations are well-suited to graphene and rGO supercapacitor electrode coatings where standard NMP-based dispersion would otherwise re-bundle.
Polyaniline and polymer-composite electrodes. A growing fraction of pseudocapacitor electrode formulations combine MXene or graphene with conductive polymers — particularly polyaniline (PANI) — that contribute both pseudocapacitance and mechanical robustness. CTI Materials holds patent coverage (U.S. Patents 10,049,783 and 11,961,630) on CNT + graphene/GO/GNP composites with polyaniline, polyaminotriazole, polyimide, polyamide, nylon, and polyester polymer matrices. For supercapacitor R&D and production buyers integrating conductive-polymer composites, the Flexiphene patented dispersion technology provides validated formulations covering these exact polymer-nanocarbon chemistries.
Cheaptubes products for supercapacitor applications
- Graphene Nanoplatelets — multiple lateral sizes and thicknesses for EDLC and hybrid electrodes
- Graphene Oxide — for in-situ rGO electrode preparation
- MXene — Ti3C2Tx powder and films for pseudocapacitor R&D
- Multi-Walled Carbon Nanotubes — for pillared MXene-CNT structures preventing restacking
- Flexiphene surfactant-stabilized dispersions — for production-scale electrode coating
For custom MXene or graphene specifications, contact Cheap Tubes Inc. directly.
Authoritative external references
- Stoller, M. D. et al. — graphene-based ultracapacitors (2008) (Nano Letters)
- Lukatskaya, M. R. et al. — MXene for high-power supercapacitors (2013) (Science)
- Ghidiu, M. et al. — clay-like MXene with high volumetric capacitance (2014) (Nature)
- Raccichini, R. et al. — graphene/GO/rGO in batteries and electrochemical capacitors, critical review (2015) (Nature Materials)
- Bonaccorso, F. et al. — graphene for batteries, supercapacitors and beyond (2016) (Nature Reviews Materials)
- Anasori, B. et al. — review of 2D MXenes for energy storage (2017) (Nature Reviews Materials)
- Simon, P. & Gogotsi, Y. — perspectives on supercapacitors (2020) (Nature Materials)
- Comprehensive review — electrochemical energy storage applications of graphene oxide including supercapacitors (2024) (Energy & Fuels)
(External links reflect representative peer-reviewed literature; no endorsement implied.)
Frequently asked questions
Graphene or MXene — which is better for supercapacitors?
It depends on the metric. For gravimetric capacitance and cost, graphene-family materials (GNP, rGO) lead. For volumetric capacitance and pseudocapacitive contribution, MXene Ti3C2Tx is best-in-class. For commercial production, graphene-based EDLCs dominate by cost; MXene leads in volumetric energy density for R&D and emerging applications.
Why doesn’t pristine graphene match its theoretical capacitance?
Restacking. Graphene sheets attract each other strongly during electrode drying — multi-layer assemblies form that lose most of the theoretical surface area. Successful electrodes use spacers (CNT intercalation, polymer linkers, hierarchical structures) to keep sheets separated and surface area accessible.
Can MXene be used in aqueous and organic electrolytes?
Yes to both, with different performance characteristics. Aqueous electrolytes deliver the highest pseudocapacitive contribution (proton-mediated surface redox). Organic electrolytes enable higher operating voltage windows but reduce pseudocapacitance. Ionic-liquid electrolytes are increasingly used for high-voltage MXene cells.
How stable is MXene in ambient air?
Pristine Ti3C2Tx MXene degrades over weeks-to-months in ambient air, particularly in humid conditions. Storage under inert atmosphere or in solvent dispersions slows degradation. Surface-functionalized MXene variants and protective coatings are active development areas for improving shelf life.
What’s the role of carbon black in supercap electrodes?
In most commercial supercapacitor electrodes, the active carbon (activated carbon, graphene, rGO) provides most of the capacitance, and 5–15 wt% carbon black is added for electrical connectivity to the current collector. Replacing carbon black with CNT or graphene in the conductive-additive role can reduce loading while improving rate capability — same percolation argument as in lithium-ion cathodes.
Continue reading
- Hub: Graphene & CNT Battery Applications
- Previous spoke: Silicon-CNT and Graphene Anodes
- Next spoke: CNT & Graphene in Li-S, Li-air, and Solid-State Batteries
About the author
Mike Foley is the founder of Cheap Tubes Inc. and CTI Materials LLC. He holds 2 granted U.S. patents (10,049,783 and 11,961,630) in carbon nanomaterial applications, with additional patents in prosecution. His patented materials were selected by NASA for the Enceladus mission as a dual-capacitance layer in ion-selective electrodes. Mike has supplied carbon nanomaterials to battery R&D and production for 21 years.

