By Mike Foley, Founder, Cheap Tubes Inc. & CTI Materials LLC — 21 years supplying carbon nanomaterials to battery R&D and production.
TL;DR
Multi-walled carbon nanotubes (MWCNT) are now the dominant conductive additive in lithium-ion cathode formulations, replacing carbon black at one-fifth the loading. Graphene and silicon-graphene composites address the cycling failure mode of high-capacity silicon anodes. Graphene, reduced graphene oxide, and MXene serve as high-surface-area electrodes in supercapacitors and pseudocapacitors. Carbon nanotube and graphene scaffolds enable next-generation lithium-sulfur and lithium-air R&D by trapping polysulfide intermediates and accommodating volumetric changes.
This guide maps each major battery application to the right carbon nanomaterial, the specifications that matter, and where to source them at research, pilot, and production scale.
Quick selection table:
| Battery problem | Material | Cheaptubes category |
|---|---|---|
| Cathode conductivity (LFP, NMC, LCO) | MWCNT 95–98%, 8–20 nm OD | Industrial CNT |
| Silicon anode expansion buffer | MWCNT 99%+, GNP, rGO | SWCNT, GNP |
| Supercapacitor electrode | GNP, rGO, MXene Ti3C2Tx | GNP, Graphene Oxide, MXene |
| Li-S polysulfide host | Porous CNT, GO scaffolds | SWCNT 99%, Graphene Oxide |
| Dispersion bottleneck (any application) | Surfactant-stabilized formulations | Flexiphene technology |
Why carbon nanomaterials matter in batteries
Three properties drive the role of carbon nanotubes and graphene in modern battery design:
- Electrical conductivity. Single-walled carbon nanotubes carry current along their axis at metallic conductivity (105–106 S/m for individual SWCNT). Even imperfect MWCNT bundles deliver 100–10,000× the conductivity of carbon black at equivalent mass loading. This translates directly into higher rate capability and lower internal resistance.
- Surface area. A single layer of graphene has a theoretical surface area of 2,630 m²/g — every carbon atom is a surface atom. Graphene oxide, reduced graphene oxide, and MXene Ti3C2Tx achieve experimentally accessible surface areas of 200–700 m²/g, which is the substrate every supercapacitor and pseudocapacitor needs to maximize stored charge.
- Mechanical strength and flexibility. Carbon nanotubes have tensile strengths 50–100× that of high-strength steel at one-sixth the density. A flexible carbon network around a brittle active material like silicon or sulfur absorbs the volumetric strain that would otherwise pulverize the electrode after a few hundred cycles.
These three properties are why every major lithium-ion cell manufacturer has either adopted or actively qualified carbon nanotubes as a conductive additive in cathodes, and why graphene composites dominate the silicon-anode patent landscape from 2018 forward.

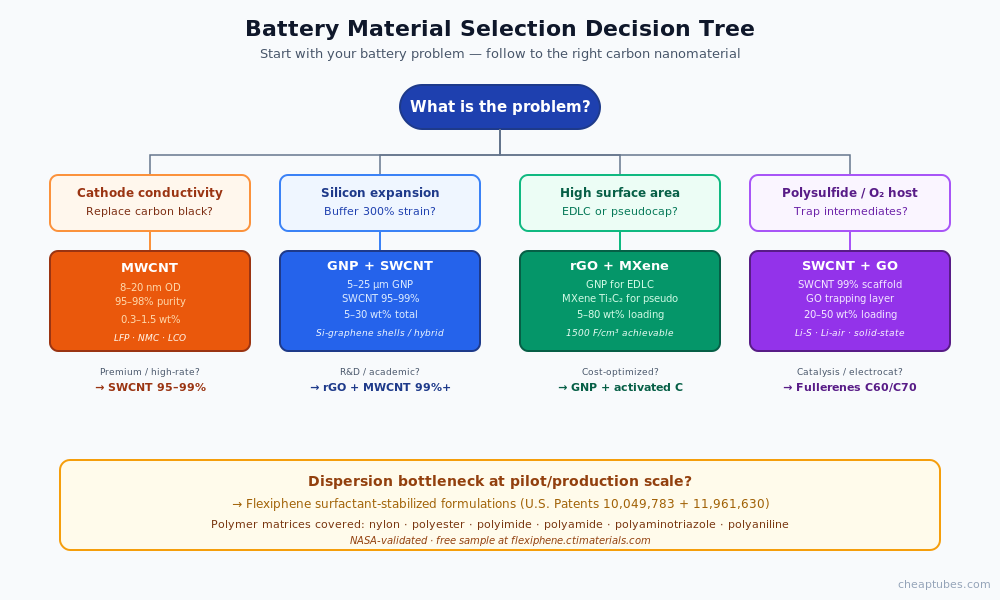
The four application categories
Battery applications for carbon nanomaterials fall into four working categories. Each has a different dominant material, a different maturity level, and a different set of specifications you need to get right.
1. Cathode conductive additive (commercial production)
MWCNT replacing carbon black in LFP, NMC, and LCO cathode formulations. Loadings of 0.3–1.5 wt% CNT achieve the same percolation network as 2–5 wt% carbon black, freeing 1.5–4 percentage points of mass for active material. This is the largest commercial CNT-in-battery application today by tonnage.
→ Read more: CNT as Cathode Conductive Additive — LFP, NMC, LCO
2. Silicon-carbon anode composites (pilot and early production)
Silicon’s theoretical capacity (4,200 mAh/g) is roughly ten times that of conventional graphite anodes, but its 300% volume expansion during lithiation pulverizes the electrode within the first few cycles. CNT and graphene matrices buffer this expansion mechanically while providing the electrical highway the disconnected silicon fragments would otherwise lose. Several major battery makers have shipped Si-C anode cells in 2024–2026.
→ Read more: Silicon-CNT & Graphene Lithium-Ion Anodes
3. Supercapacitor and pseudocapacitor electrodes (commercial)
Graphene nanoplatelets, reduced graphene oxide, and MXene serve as high-surface-area conductive substrates in EDLCs and pseudocapacitors. MXene contributes both double-layer capacitance and surface-redox pseudocapacitance, achieving volumetric capacitances above 1,500 F/cm³ in optimized films. Commercial supercapacitor manufacturers have shipped graphene-based devices since 2016.
→ Read more: Graphene & MXene Supercapacitor Electrodes
4. Next-generation chemistries: Li-S, Li-air, solid-state (R&D)
Carbon nanotube and graphene scaffolds are the dominant cathode host architecture in lithium-sulfur cell research, where porous carbon traps soluble polysulfide intermediates that would otherwise shuttle between electrodes. Lithium-air and solid-state battery R&D similarly leverage carbon nanostructures for catalysis and interfacial conduction. Commercial deployment is 2027+ for most of these chemistries.
→ Read more: CNT & Graphene in Li-S, Li-air, and Solid-State Batteries
Materials × applications: which spec for which problem
The same nanomaterial label hides a wide range of grades. A 95% MWCNT optimized for industrial cathode slurry is different from a 99.9% MWCNT used in academic Si-anode research. Here is the practical selection matrix:
| Material | Cathode additive | Si anode | Supercapacitor | Li-S / next-gen | Typical loading | Notes |
|---|---|---|---|---|---|---|
| SWCNT 95% | Specialty | ✓ | – | ✓ | 0.05–0.5 wt% | Best aspect ratio, premium price |
| SWCNT 99% | – | ✓ | – | ✓ (R&D) | 0.05–0.5 wt% | R&D-grade, excellent for paper publication |
| MWCNT 8–20 nm, 95% | ✓✓✓ | – | – | – | 0.3–1.5 wt% | Cathode workhorse — best price/performance |
| MWCNT 8–20 nm, 98% | ✓✓ | ✓ | – | – | 0.3–1.5 wt% | Cleaner cathode, anode-friendly |
| MWCNT 99.9% | – | ✓ | – | ✓ | 0.5–2 wt% | Academic R&D, ultra-low metal residual |
| GNP (graphene nanoplatelets) | – | ✓ | ✓✓ | ✓ | 1–10 wt% | Anode buffer, supercap conductive backbone |
| GO (graphene oxide) | – | – | ✓ | ✓✓ | 1–10 wt% | Polysulfide trapping via oxygen groups |
| rGO (reduced graphene oxide) | – | ✓ | ✓✓ | ✓ | 1–10 wt% | Conductivity restored, defect-stabilized |
| MXene Ti3C2Tx | – | – | ✓✓✓ | – | 5–80 wt% (electrode) | Pseudocapacitive, premium price |
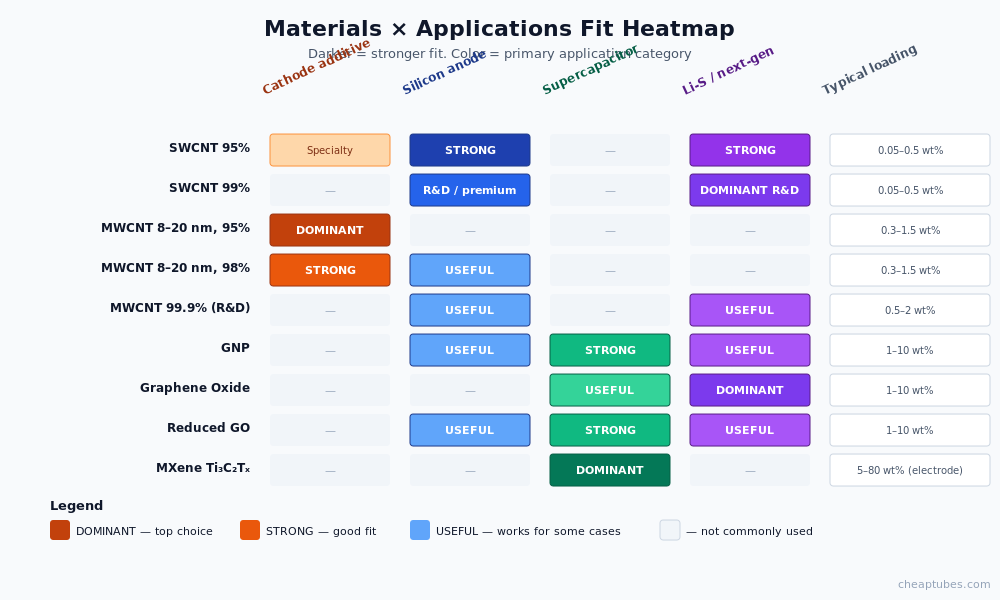
The most common selection mistake is pulling the highest-purity or highest-spec material when the application doesn’t require it. A 99.9% MWCNT in a cathode slurry adds cost without measurable performance benefit — the binder, electrolyte interface, and cell-level engineering will dominate any gain. Save the high-purity grades for fundamental R&D where metal residuals would interfere with measurement.
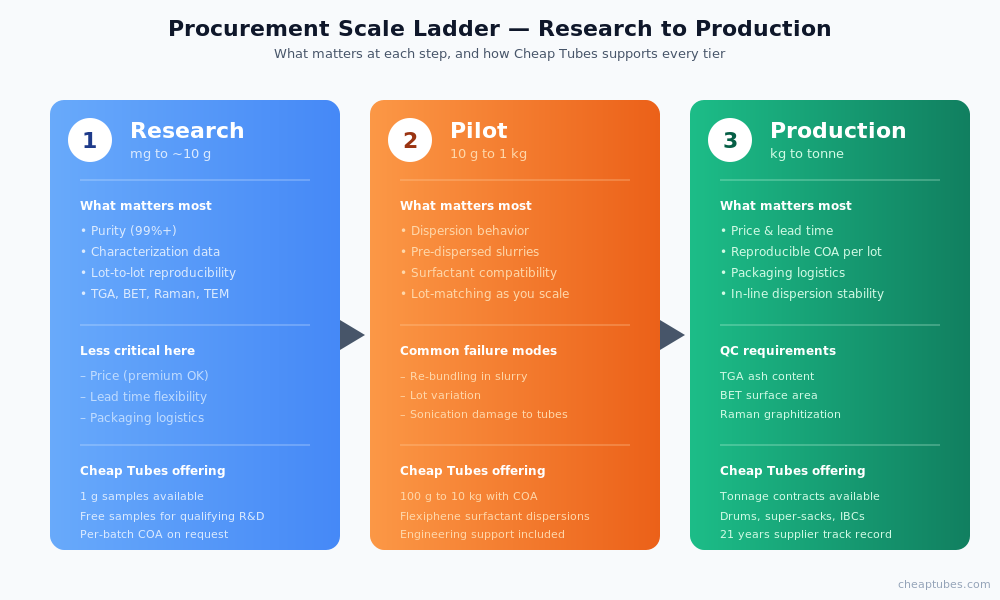
Procurement realities — research, pilot, and production scale
Most published battery papers use grams to tens of grams of carbon material. Most production cells use kilograms to tonnes per year. The supply chain considerations differ at every step.
Research scale (mg to ~10 g): Purity, characterization data, and lot-to-lot reproducibility matter more than price. Buy what your method requires, document the lot number, and accept the per-gram premium.
Pilot scale (10 g to 1 kg): Dispersion behavior becomes the dominant variable. Two MWCNT lots with the same nominal spec can disperse very differently in NMP, water, or your specific binder system. Source from a supplier who will provide pre-dispersed slurries or surfactant-stabilized formulations and who can talk through dispersion strategy. This is where tools like the Flexiphene patented surfactant technology earn their keep — the dispersion problem alone wastes more pilot batches than any other failure mode.
Production scale (kg to tonne): Price, lead time, COA reproducibility, and packaging logistics dominate. Lock in a supplier who can deliver in your packaging spec (drum, super-sack, IBC), with a reproducible COA per lot, and who has the capacity to scale with you. The right supplier should be running quality control on every lot — TGA for ash content, BET for surface area, Raman for graphitization quality, TEM for diameter and length distribution.
Cheap Tubes Inc. covers all three scales. Researchers can buy 1 g samples; pilot teams can buy 100 g–10 kg with characterization data; production buyers can negotiate tonnage contracts with consistent COA and packaging.
Working with hard-to-disperse materials
Single-walled CNT, the highest-aspect-ratio multi-walled CNT, and many graphene grades are notoriously difficult to disperse uniformly. Bundling, agglomeration, and re-aggregation in slurries are the most common failure modes in battery R&D and the most expensive failure mode in production scale-up.
Three approaches to manage dispersion in battery applications:
- Aggressive mechanical processing — high-shear mixing, ultrasonication, three-roll milling. Effective but consumes energy, can damage long tubes (cutting them shorter and reducing aspect ratio), and rarely scales cleanly.
- Functionalization — chemical attachment of -COOH, -OH, or -NH2 groups to the carbon surface improves polar-solvent compatibility. Useful for water-based systems but can degrade conductivity if over-functionalized. Cheap Tubes’ functionalized MWCNT category covers the major functional group options.
- Surfactant-stabilized formulations — non-covalent dispersion using engineered surfactants. Preserves the carbon’s electrical and mechanical properties while delivering a stable dispersion that can be loaded into cathode slurries, anode pastes, or supercapacitor electrode coatings without re-bundling. The Flexiphene patented surfactant system developed by CTI Materials addresses this category specifically — NASA validation and 100× lower interfacial resistance vs comparable non-stabilized formulations make it the strongest commercial option for battery dispersion at scale. Free sample request available at the link.
For lab batches, options 1 and 2 typically suffice. For pilot or production scale, option 3 is the only approach that consistently delivers reproducible electrode coatings without per-batch re-engineering.
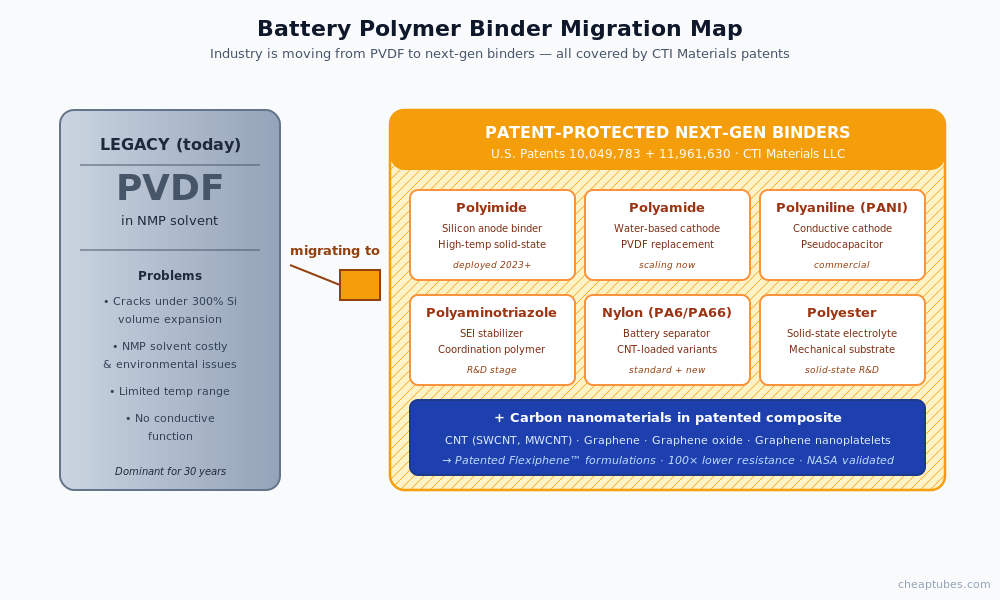
Patent-protected polymer-nanocarbon composite formulations
The challenge in moving battery composites from R&D to production is not the carbon material itself — it is the polymer matrix that must hold the carbon dispersed, conduct ions or electrons, and survive thousands of charge cycles without degradation. The battery industry is migrating away from legacy PVDF binders toward a defined set of polymer systems: polyimide for silicon anodes, polyamide and water-soluble polyamide variants for cathodes, polyaniline and polyaminotriazole as conductive coatings, nylon for separators, and polyester-based films for solid-state architectures.
CTI Materials holds patent coverage (U.S. Patents 10,049,783 and 11,961,630) on polymer-nanocarbon composite formulations specifically combining CNT, graphene, graphene oxide, and graphene nanoplatelets with these exact polymers — nylon, polyester, polyimide, polyamide, polyaminotriazole, and polyaniline. The Flexiphene patented surfactant technology delivers stable dispersions in each of these polymer matrices, with NASA-validated performance data (100× lower interfacial resistance, 96× higher capacitance, +19% polymer strength at 1 wt% loading).
For battery R&D and production teams, this means a single sourcing relationship covers both the carbon nanomaterials and the patent-protected polymer-composite formulations — eliminating the integration risk of stitching together suppliers across the composite stack. For licensees and OEMs developing proprietary chemistries, the patent portfolio provides freedom-to-operate options for the polymer systems most likely to scale in lithium-ion, silicon-anode, supercapacitor, and solid-state battery production.
Request a Flexiphene technical sample →
Authoritative external references
- Yoo, E. et al. — graphene nanosheets as LIB anode active material delivering 540 mAh/g vs graphite’s 372 mAh/g (foundational, 2008) (Nano Letters)
- Chou, S.-L. et al. — first Si/graphene composite anode demonstrating 1168 mAh/g over 30 cycles (2010) (Electrochem. Commun.)
- Ji, L. et al. — graphene oxide as sulfur immobilizer in high-performance Li/S cells, foundational paper for GO trapping (2011) (JACS)
- Lukatskaya, M. R. et al. — MXene for high-power supercapacitors (2013) (Science)
- Raccichini, R. et al. — comprehensive review of graphene/GO/rGO in batteries and electrochemical capacitors (2015) (Nature Materials)
- Bonaccorso, F. et al. — graphene for batteries, supercapacitors and beyond (2016) (Nature Reviews Materials)
- Tang, J., Pang, J. & Wang, J. — covalent 3D CNT@rGO nano-hybrid for high-efficiency LiFePO4 cathodes (2025) (Advanced Science)
- Tong, X. et al. — comprehensive review of CNT applications in lithium-ion batteries (2025) (Carbon Energy)
- DOE Office of Electricity — Energy Storage Grand Challenge Roadmap
(External links reflect representative peer-reviewed literature; no endorsement implied.)
Frequently asked questions
Why use carbon nanotubes instead of carbon black in cathodes?
Carbon nanotubes form a percolation network at far lower mass loading than spherical carbon black particles because of their high aspect ratio. Typical industrial cathodes use 2–5 wt% carbon black; equivalent percolation is achieved with 0.3–1.5 wt% MWCNT. The mass freed by lower additive loading is added back as active material, raising cell-level energy density by 1–4%.
What loading of MWCNT is typical in LFP cathodes?
Most production LFP formulations use 0.5–1.0 wt% MWCNT as the sole conductive additive, or 0.2–0.5 wt% MWCNT combined with a reduced carbon-black fraction. The exact loading depends on cathode coating thickness, target rate capability, and binder system. Higher MWCNT loadings improve rate capability but at a cost premium.
Can SWCNT replace MWCNT for cathode conductive additive?
Technically yes — SWCNT achieves percolation at even lower loading (0.05–0.2 wt%) — but the per-gram cost difference (5–20× higher) means most production cells stay with MWCNT. SWCNT in cathodes appears in premium niche products and high-rate cells where the conductivity gain justifies the cost.
What’s the role of graphene in silicon anodes?
Graphene serves three functions simultaneously in silicon anode composites: a flexible mechanical buffer that absorbs the 300% volume expansion of silicon during lithiation; an electrical conductor that maintains contact with silicon fragments after they crack; and a structural matrix that limits the SEI growth which would otherwise consume electrolyte cycle after cycle.
Why MXene for supercapacitors and not lithium-ion batteries?
MXene’s high pseudocapacitive contribution and excellent in-plane conductivity are well-matched to charge/discharge timescales of seconds (supercapacitors). At lithium-ion timescales (minutes to hours of charge/discharge), MXene’s low gravimetric capacity vs lithium intercalation materials means it can’t compete with graphite or silicon as a primary anode material. MXene’s role in lithium chemistry is mostly as a conductive scaffold or interlayer rather than the active material.
Are functionalized CNTs needed for batteries?
For most cathode and anode applications, pristine MWCNT or SWCNT performs better than functionalized variants — the functional groups disrupt the conjugated π-network that delivers the electrical conductivity benefit. Functionalized CNT is most useful when dispersion in a polar solvent (especially water) is a hard requirement and you cannot use a surfactant approach. For polysulfide trapping in Li-S, the oxygen groups on graphene oxide are an exception — they actively bind polysulfide species, which is exactly the desired behavior.
What dispersion methods work best for battery slurries?
Lab scale: ultrasonic horn or bath sonication for short dispersions (5–30 minutes), with mechanical stirring during slurry homogenization. Pilot scale: high-shear mixers (e.g., IKA, Silverson), three-roll mill for high-viscosity slurries, planetary ball mills for solid-state cathode preparation. Production scale: in-line high-shear mixing combined with surfactant-stabilized pre-dispersions to skip the most variable steps.
How to scale from lab grams to production kilograms?
Three rules: (1) lock the specification — diameter range, length range, purity, and ash content must stay within tight bounds across orders; (2) use the same dispersion approach at every scale, even if mechanical method changes — surfactant-stabilized pre-dispersions are the most scale-stable; (3) validate at each step — do not skip from 100 g to 10 kg without an intermediate batch. Cheap Tubes provides matched lots at each scale to minimize lot-to-lot variation as you scale.
Get started
For specific battery application categories, start with one of the dedicated guides above. For materials selection, the most commonly used categories for battery work are:
- Industrial-Grade Carbon Nanotubes — production-scale MWCNT for cathode and anode work
- Single-Walled Carbon Nanotubes — high-purity SWCNT for premium and R&D applications
- Graphene Nanoplatelets — anode and supercapacitor work
- Graphene Oxide — polysulfide trapping, supercapacitor films
- MXene — pseudocapacitor electrodes
- Functionalized Carbon Nanotubes — polar-solvent compatible variants
For dispersion-limited applications at pilot or production scale, request a sample of Flexiphene surfactant-stabilized nanocarbon dispersions — NASA-validated, U.S. patented, free sample request available.
For custom formulations, custom MWCNT specifications, or tonnage supply discussions, contact Cheap Tubes Inc. directly.
About the author
Mike Foley is the founder of Cheap Tubes Inc. and CTI Materials LLC. He holds 2 granted U.S. patents (10,049,783 and 11,961,630) in carbon nanomaterial applications, with additional patents in prosecution. His patented materials were selected by NASA for the Enceladus mission as a dual-capacitance layer in ion-selective electrodes. Mike has supplied carbon nanomaterials to battery R&D and production for 21 years.

