By Mike Foley, Founder, Cheap Tubes Inc. & CTI Materials LLC.
Part of the Graphene & CNT Battery Applications hub.
TL;DR
Lithium-sulfur (Li-S), lithium-air (Li-O2), and solid-state battery chemistries promise 2–10× the energy density of conventional lithium-ion, but each faces a distinct materials challenge that carbon nanotubes and graphene help solve. In Li-S, porous CNT and graphene hosts trap soluble polysulfide intermediates that would otherwise shuttle between electrodes and destroy capacity. In Li-air, CNT/graphene scaffolds catalyze oxygen reduction and accommodate Li2O2 deposition. In solid-state cells, graphene at the electrolyte/electrode interface improves wettability and reduces interfacial resistance. This page covers R&D-grade carbon material selection and procurement for these next-generation chemistries.
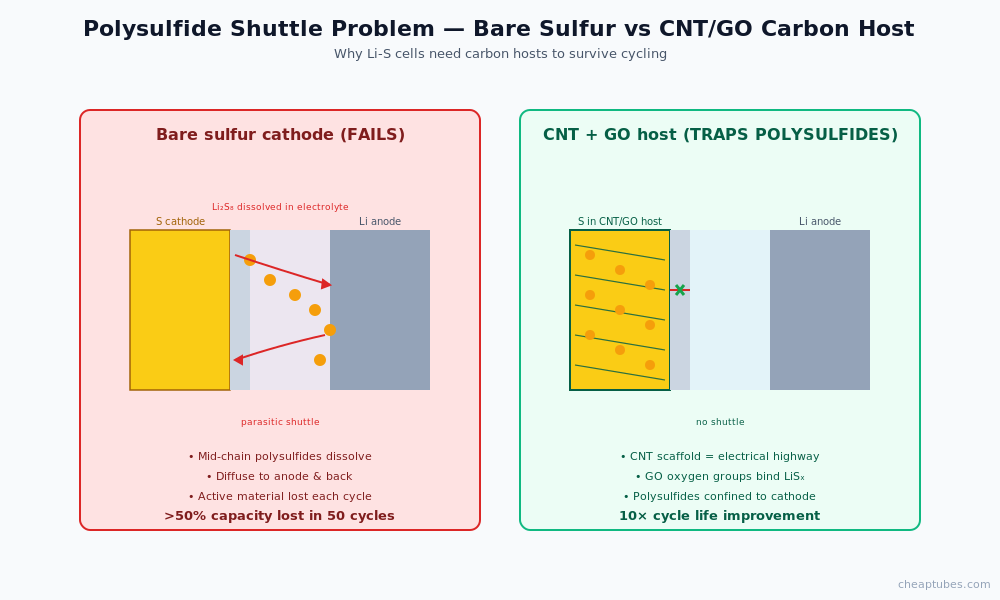
The polysulfide shuttle problem in Li-S
Lithium-sulfur cells have a theoretical specific energy of approximately 2,600 Wh/kg — roughly 5× that of conventional lithium-ion. The cathode is elemental sulfur, which is abundant, cheap, and lightweight. The barrier to commercialization is the polysulfide shuttle effect:
During discharge, sulfur progressively combines with lithium through intermediate lithium polysulfides (Li2S8, Li2S6, Li2S4, Li2S2) before reaching the final Li2S product. The mid-chain polysulfides (Li2S8, Li2S6, Li2S4) are soluble in the conventional liquid electrolyte. Once dissolved, they diffuse across the separator to the lithium anode, react with lithium metal directly, and either form an insulating layer on the anode or return to the cathode as longer-chain polysulfides — a parasitic shuttle that consumes active material and lithium with every cycle.
The result: pristine sulfur cathodes lose more than 50% of capacity within the first 50 cycles. The shuttle effect, not the sulfur itself, is the bottleneck.
Carbon nanostructures address this by trapping polysulfides physically and chemically before they can dissolve and diffuse.
Carbon hosts for sulfur cathodes
Three carbon-host architectures dominate published Li-S cathode work:
1. Mesoporous CNT scaffolds. Sulfur is melt-infiltrated into the pores of a CNT network. The high-aspect-ratio CNT framework provides electrical connectivity to the sulfur and physically confines the polysulfide intermediates within the pore network. Best-performing implementations use SWCNT 99% or MWCNT 99.9% with engineered pore sizes 2–10 nm.
2. Graphene oxide trapping layers. GO has oxygen functional groups (-OH, -COOH, epoxy) that interact chemically with polysulfide species. Polysulfides bond to the oxygen groups, preventing dissolution. GO is used both as the sulfur host and as a separator interlayer that catches escaping polysulfides. Loadings 20–60 wt% GO in the cathode are common in R&D formulations.
3. Hierarchical CNT/graphene composite hosts. Combining CNT scaffolding (electrical highway, mechanical support) with graphene oxide trapping (chemical polysulfide binding) yields composite cathodes with both mechanisms operating simultaneously. Most published high-cycle-life Li-S cells in 2024–2026 use this hybrid approach.
Material specifications for Li-S cathode hosts:
| Material | Function | Loading | Notes |
|---|---|---|---|
| SWCNT 99% | Conductive scaffold for sulfur | 5–15 wt% | High aspect ratio, low ash |
| MWCNT 99.9% | Mesoporous host | 10–30 wt% | Best for pore-engineering studies |
| Graphene oxide | Chemical polysulfide trap | 20–50 wt% | Oxygen functional groups bind LiSx |
| Reduced GO | Conductive backbone + trap | 20–40 wt% | Partial defect retention preserves binding |
The high carbon loading (often 30–50 wt% in academic R&D) is the trade-off for cycle life — sulfur utilization drops as carbon mass goes up. Production-feasibility work targets 20% carbon while maintaining shuttle suppression.
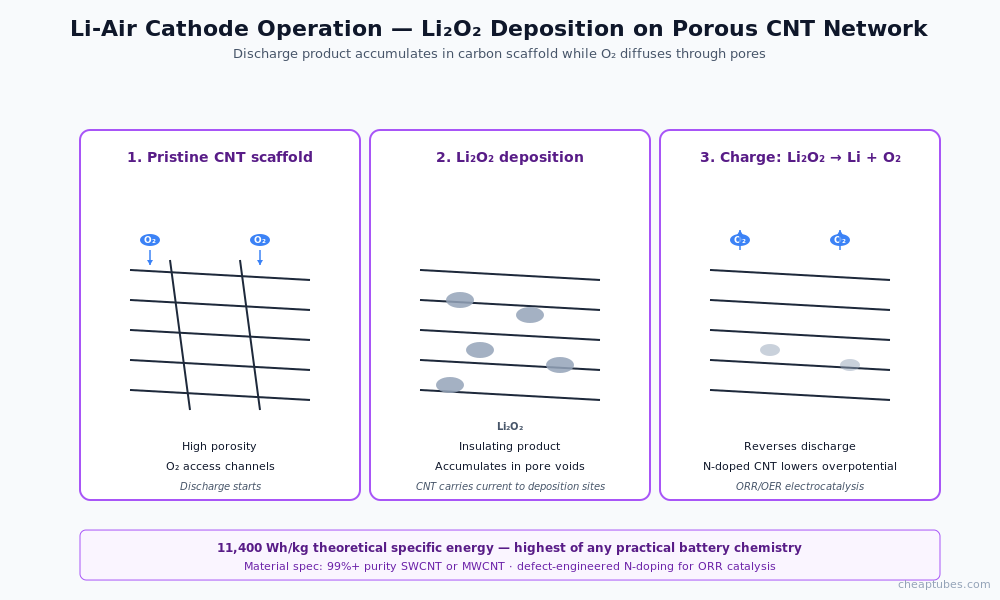
Lithium-air cathodes
Lithium-air (Li-O2) cells have a theoretical specific energy near 11,400 Wh/kg using oxygen from ambient air as the cathode reactant — the highest of any practical battery chemistry. The cathode reaction forms lithium peroxide (Li2O2), an insulating solid that must accumulate on a porous conductive scaffold during discharge and be decomposed back to lithium and oxygen during charge.
Carbon scaffolds are the dominant cathode architecture in Li-air R&D for three reasons:
- Porosity for Li2O2 accommodation. Discharge product Li2O2 is an insulating solid that must accumulate without blocking oxygen access. Porous CNT or graphene networks provide the void space for deposition while maintaining oxygen diffusion paths.
- Catalytic activity for oxygen reduction. Defective graphene and N-doped CNT exhibit measurable electrocatalytic activity for the oxygen reduction reaction (ORR), reducing the cathode overpotential during discharge.
- Conductive backbone for insulating discharge products. Once Li2O2 accumulates, only the carbon scaffold can carry current; the discharge product itself is insulating. CNT/graphene scaffolds maintain electrical connectivity to the deposition sites.
Material specifications converge on similar grades to Li-S work: high-purity SWCNT or MWCNT (99%+), and high-quality graphene oxide or rGO. The lower commercial volume of Li-air R&D vs Li-S means specifications are typically academic-grade rather than industrial production.
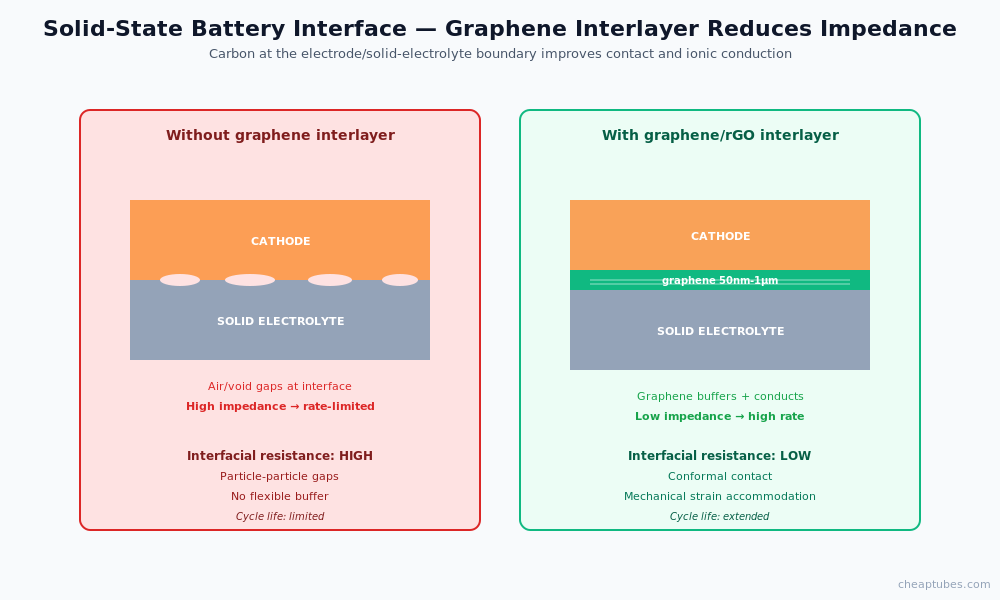
Solid-state battery interfaces
Solid-state batteries replace the liquid electrolyte with a solid-electrolyte layer (sulfide-based, oxide-based, or polymer-based). The fundamental advantage is safety and high-voltage compatibility; the fundamental challenge is the electrode-electrolyte interface, where poor contact creates high interfacial resistance and cycle-life problems.
Graphene-family materials appear in solid-state battery research in two roles:
- Interfacial conductive layer. A thin graphene or rGO film at the electrode/solid-electrolyte interface improves electrical and ionic contact, reducing the impedance ladder that limits high-rate cycling. Layer thicknesses of 50 nm to 1 µm are typical.
- Cathode composite for sulfide solid electrolytes. Sulfide solid electrolytes are sensitive to oxidation at high-voltage cathodes. CNT and graphene composites with the cathode active material can buffer the interface and extend the stable voltage window.
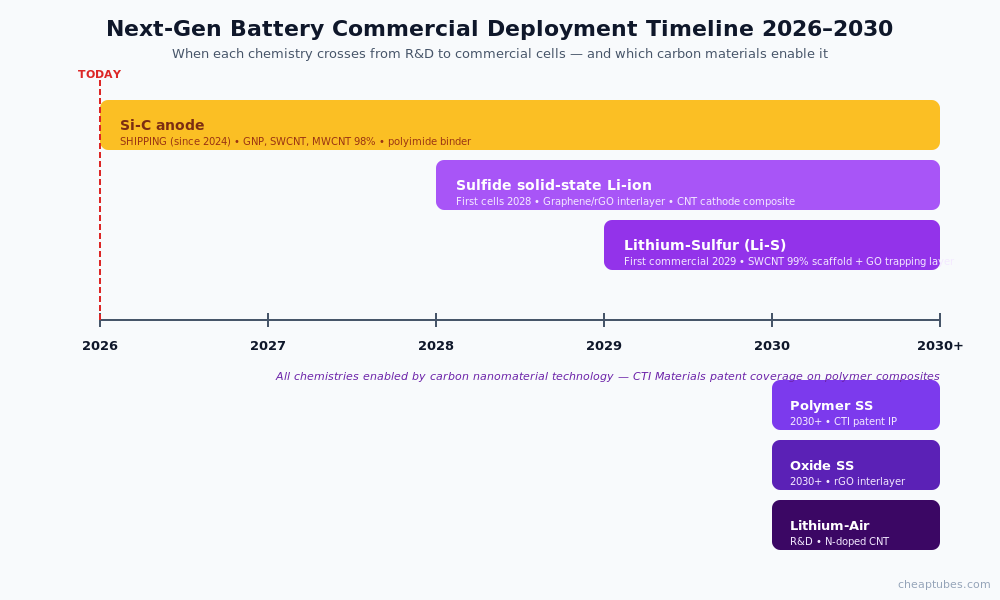
Solid-state work is currently the smallest commercial volume of the four next-gen chemistries but the largest R&D pipeline, with multiple commercial deployment targets in 2027–2030.
Polymer electrolytes and CNT/graphene interfaces
Solid-state batteries using polymer-electrolyte architectures rely on a different set of polymer chemistries than conventional lithium-ion: polyethylene oxide (PEO) and its successors, polyimide for high-temperature operation, and polyaminotriazole-family coordination polymers for high-conductivity solid electrolytes under R&D. The polymer-electrolyte/electrode interface is one of the dominant performance bottlenecks for solid-state cells, and CNT/graphene composite interlayers are an active area of research for improving this interface.
CTI Materials’ Flexiphene patent portfolio (U.S. Patents 10,049,783 and 11,961,630) covers CNT + graphene/GO/GNP composites with the exact polymer matrices appearing in solid-state battery R&D — polyimide, polyamide, polyaniline, polyaminotriazole, nylon, and polyester. For research groups and battery-startup R&D teams working on polymer-electrolyte solid-state cells, this means a single technology source covers both the carbon nanomaterial and the patent-protected polymer-composite formulation chemistry. The Flexiphene patented system provides NASA-validated dispersion performance in these exact polymer systems.
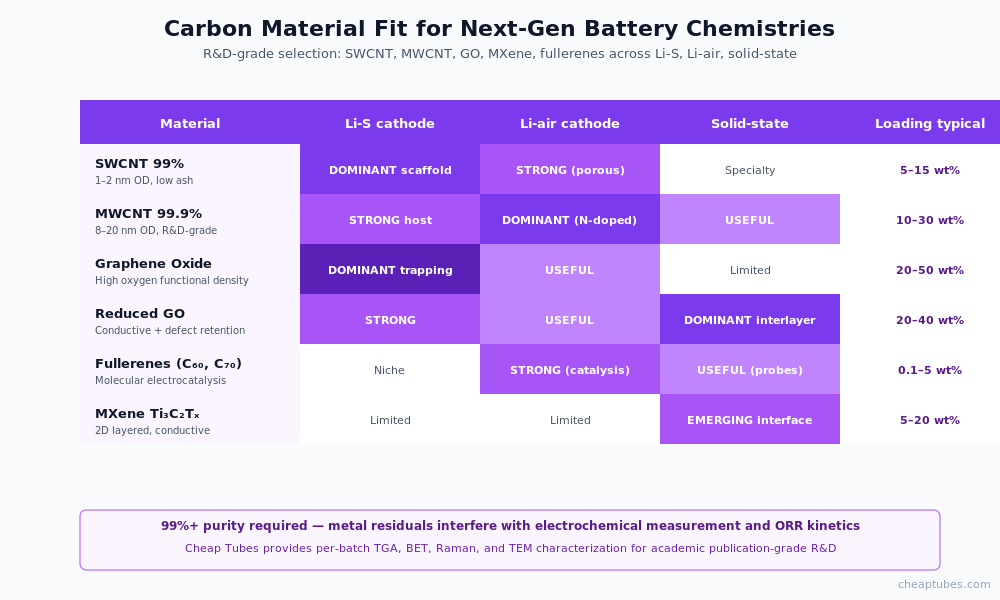
Material specs for R&D-grade work
Next-gen battery R&D requires the highest-purity carbon grades for two reasons. First, metal residuals (catalyst remnants from CVD synthesis) interfere with electrochemical measurement — they catalyze unwanted side reactions, mask intrinsic capacity, and complicate interpretation of published results. Second, the small batch sizes typical of academic R&D allow premium-grade procurement without prohibitive cost.
Recommended grades:
- SWCNT 99% — primary scaffold material; lowest ash content available in catalog grades; available in 0.5–10 g batches at Cheap Tubes
- MWCNT 99.9% — high-purity multi-walled CNT for pore engineering and percolation studies
- Graphene Oxide — high oxygen functional density for polysulfide trapping
- Single-Layer Graphene Oxide — monolayer GO for trapping-layer R&D
- Fullerenes (C60, C70) — molecular carbon for electrocatalysis and interface studies
For specific batch characterization (TGA ash content, BET surface area, Raman, TEM imaging), Cheap Tubes provides per-batch characterization data on request — important for reproducible academic publication.
Cheaptubes products for next-gen battery R&D
- Single-Walled Carbon Nanotubes — 95–99.9% purity grades suitable for Li-S, Li-air, and solid-state R&D
- Multi-Walled Carbon Nanotubes — 98–99.9% purity options for pore-engineering studies
- Graphene Oxide — including single-layer GO for polysulfide trapping research
- Fullerenes (C60, C70) — 95–99.9% purity grades for electrocatalysis and molecular electronics work
- MXene — emerging role in solid-state battery interfaces
For academic R&D requiring documented characterization on each batch, contact Cheap Tubes Inc. directly. Free samples are available for qualifying R&D projects.
Authoritative external references
- Ji, L., Rao, M., Zheng, H. et al. — graphene oxide as sulfur immobilizer in high-performance Li/S cells, foundational paper for GO trapping (2011) (JACS)
- Ji, X. et al. — sulfur-graphene oxide nanosheet cathode for Li-S (2011) (Nature Communications)
- Manthiram, A. et al. — review of Li-S battery cathode design (2014) (Chemical Reviews)
- Janek, J. & Zeier, W. G. — solid-state battery roadmap (2016) (Nature Energy)
- Liu, T. et al. — review of Li-air battery electrocatalysts (2017) (Chemical Society Reviews)
- Li, X. et al. — self-catalyzed Co4N + N-doped CNTs as bifunctional cathode for flexible Li-air batteries (2024) (ACS Nano)
- Graphene and CNT-based hybrid nanocomposite review covering Li-S, Li-air, supercapacitors, fuel cells (2025) (Synthetic Metals)
- DOE Office of Electricity — Energy Storage Grand Challenge Roadmap
(External links reflect representative peer-reviewed literature; no endorsement implied.)
Frequently asked questions
Why does Li-S need a carbon host — why not just use sulfur directly?
Pure sulfur cathodes lose more than 50% of capacity within 50 cycles because polysulfide intermediates dissolve in the electrolyte and shuttle to the anode. The carbon host traps polysulfides physically (in pores) and chemically (oxygen groups bind LiSx), keeping the sulfur active material in the cathode and extending cycle life by 10× or more.
Is GO or rGO better for polysulfide trapping?
GO has more oxygen functional groups and stronger chemical binding to polysulfides — better trapping per gram. rGO has higher electrical conductivity — better current collection. Most high-performance Li-S cathodes use a combination: GO for the trapping interlayer or external layer; rGO or CNT for the conductive scaffold.
What CNT purity is needed for Li-air R&D?
99%+ at minimum. Metal residuals from synthesis catalysts interfere with oxygen reduction kinetics — they can either catalyze unwanted side reactions or dominate the measured cathode performance. For peer-reviewed publication, 99.5–99.9% MWCNT or 99% SWCNT is standard.
When will solid-state batteries reach commercial deployment?
Most credible roadmaps target initial commercial deployment in 2027–2029, with broader market entry through 2030. Sulfide solid-electrolyte cells lead the production timeline; oxide and polymer solid-electrolyte systems target later windows. Carbon-graphene interface engineering is a critical enabler for high-rate solid-state cells.
Are fullerenes used in any commercial battery?
Not currently in commercial production. Fullerene research in batteries focuses on electrocatalysis (oxygen reduction in Li-air), interface engineering (solid-state cathodes), and as a molecular probe for studying lithium-carbon interactions. Commercial deployment requires step-changes in either fullerene synthesis cost or in a battery chemistry where fullerenes are uniquely enabling — neither has yet emerged.
Continue reading
- Hub: Graphene & CNT Battery Applications — overview of all four application categories
- Previous spokes:
- CNT Cathode Conductive Additive
- Silicon-CNT and Graphene Anodes
- Graphene & MXene Supercapacitor Electrodes
- Buying guides: SWCNT, MWCNT, GNP
About the author
Mike Foley is the founder of Cheap Tubes Inc. and CTI Materials LLC. He holds 2 granted U.S. patents (10,049,783 and 11,961,630) in carbon nanomaterial applications, with additional patents in prosecution. His patented materials were selected by NASA for the Enceladus mission as a dual-capacitance layer in ion-selective electrodes. Mike has supplied carbon nanomaterials to battery R&D and production for 21 years.

