By Mike Foley, Founder, Cheap Tubes Inc. & CTI Materials LLC — 21 years supplying research-grade carbon nanotubes to scientists, engineers, and industrial customers.
TL;DR
Carbon nanotubes (CNTs) are cylindrical molecules built entirely from carbon atoms arranged in hexagonal lattices and rolled into seamless tubes 0.7–50 nanometers in diameter and ranging from sub-micrometer to centimeters in length. Discovered in 1991 by Sumio Iijima at NEC, they are among the strongest materials known — tensile strengths over 50× that of high-strength steel at one-sixth the density — and they conduct electricity along their axis at metallic densities while exhibiting axial thermal conductivities higher than copper or diamond.
Carbon nanotubes are classified by wall count: single-walled (SWCNT) — one graphene cylinder, double-walled (DWCNT) — two concentric cylinders, and multi-walled (MWCNT) — three or more nested cylinders. This combination of mechanical strength, electrical and thermal conductivity, and one-dimensional geometry makes carbon nanotubes the active component in lithium-ion battery cathodes, polymer composites, EMI shielding films, supercapacitor electrodes, sensors, and emerging electronics.
Quick selection table:
| What you need | Best CNT type | Where to learn more |
|---|---|---|
| Mechanical strength + light weight in a composite | SWCNT or DWCNT, 1–4 nm OD | SWCNT Buying Guide |
| Conductivity + cost-effective scale (cathode additive, EMI) | MWCNT 8–20 nm, 95–98% | MWCNT Buying Guide |
| Academic R&D / publication-grade | MWCNT 99.9% or SWCNT 99% | Cheap Tubes catalog |
| Water-based dispersion or polar binder system | Functionalized MWCNT (-COOH, -OH, -NH2) | Functionalized CNT |
Discovery and molecular structure
The widely-cited discovery of carbon nanotubes belongs to Sumio Iijima, who in 1991 published electron-microscopy observations of “helical microtubules of graphitic carbon” in Nature while at NEC’s Tsukuba laboratory. Earlier observations of similar structures appeared in Russian and Western literature from the 1950s onward, but Iijima’s paper established the modern field by combining clear imaging, structural interpretation, and reproducible synthesis.
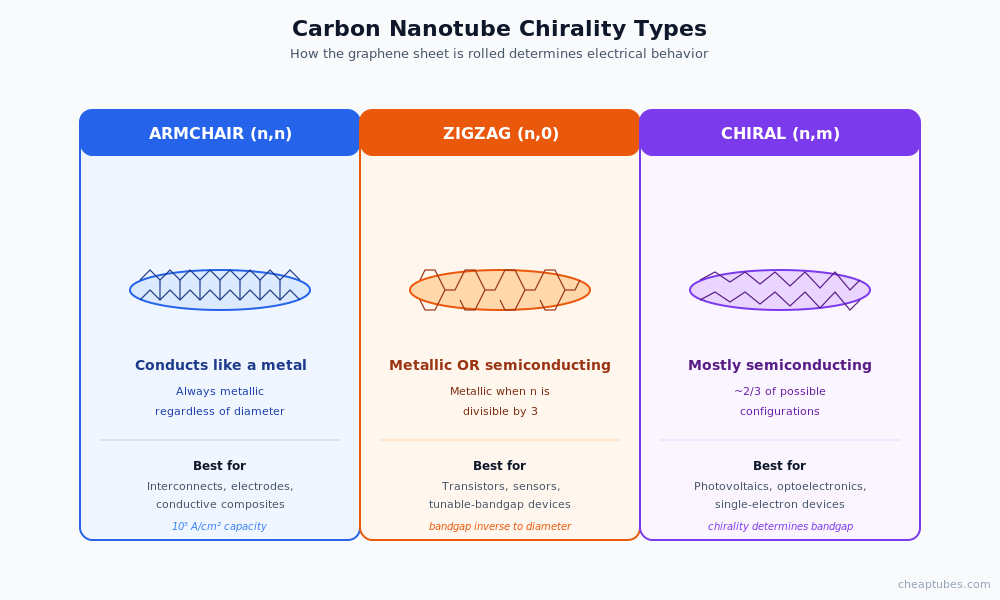
At the atomic scale, a carbon nanotube is a single sheet of graphene — a one-atom-thick hexagonal mesh of sp² carbon — rolled into a seamless cylinder. The way the graphene sheet is rolled defines the nanotube’s “chirality,” which in turn controls its electrical behavior. Three chirality types are recognized:
- Armchair nanotubes have a specific axial symmetry that makes them metallic conductors regardless of diameter.
- Zigzag nanotubes are metallic only when the chiral index (n,0) has n divisible by three; otherwise they are semiconducting with diameter-dependent bandgap.
- Chiral nanotubes follow similar rules — most are semiconducting, a minority are metallic.
The two ends of a carbon nanotube can be open (terminated in dangling bonds, typically passivated by hydrogen, oxygen, or hydroxyl groups) or capped with hemispherical fullerene-like structures. Defects in the wall — Stone-Wales rearrangements, vacancies, sp³ centers — modify mechanical and electrical properties and provide useful chemistry for functionalization.
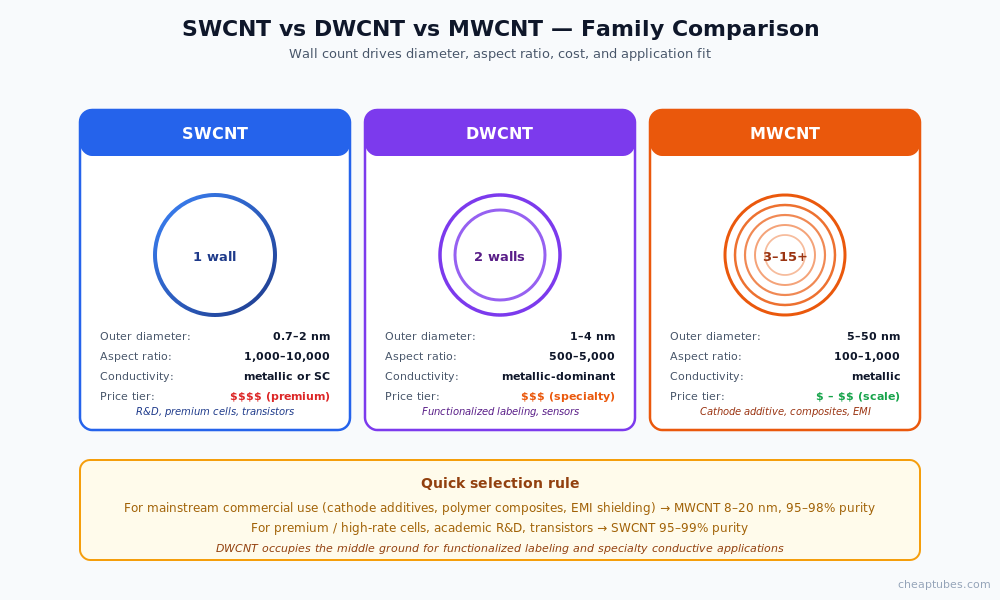
The three families: SWCNT, DWCNT, MWCNT
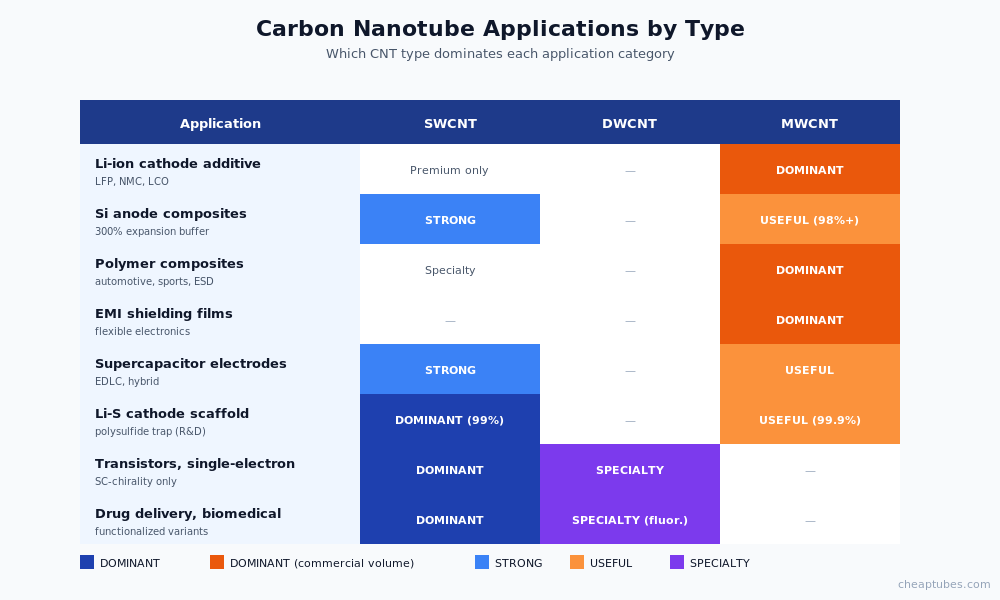
Carbon nanotubes are classified by wall count, and the differences matter enormously for both performance and price.
Single-walled carbon nanotubes (SWCNT)
A single graphene cylinder, typically 0.7–2 nanometers in outer diameter. SWCNT exhibits the highest theoretical mechanical strength (Young’s modulus ~1 TPa, tensile strength up to 100 GPa) and the highest current-carrying capacity (109 A/cm² — orders of magnitude beyond copper). Production yield is the lowest of the three families, which translates into the highest per-gram cost — typically 5–20× the price of equivalent MWCNT. SWCNT applications include academic research, premium high-rate battery cells, transparent conductive films, single-electron transistors, and sensors where the highest conductivity-to-mass ratio is essential.
Double-walled carbon nanotubes (DWCNT)
Two concentric graphene cylinders, 1–4 nanometers outer diameter. DWCNT occupies a useful middle ground: nearly the conductivity and aspect ratio of SWCNT, but with the additional mechanical robustness and chemical-handling resilience of having two walls. The outer wall can be functionalized for solubility or matrix compatibility while the inner wall preserves electronic properties. Applications include functionalized fluorescent labeling, reinforced composites, and emerging electronics where SWCNT is too fragile or expensive.
Multi-walled carbon nanotubes (MWCNT)
Three to fifteen or more concentric graphene cylinders, 5–50 nanometers outer diameter typically (specialty grades reach 100 nm). Two morphological sub-types exist: the Russian-doll structure (concentric continuous cylinders) and the bamboo structure (compartmented internal walls). MWCNT is the workhorse for tonnage commercial applications — lithium-ion cathode conductive additive, polymer composites, EMI shielding, conductive plastics — because it offers approximately metallic conductivity at one-fifth to one-twentieth the per-gram cost of SWCNT.
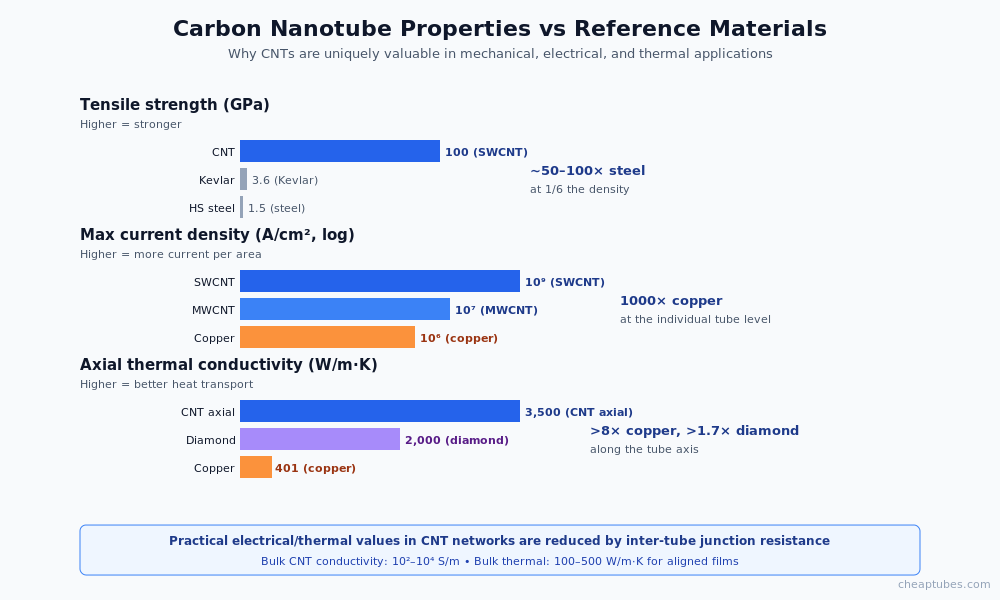
Properties that drive applications
Three properties make carbon nanotubes uniquely valuable across many engineering disciplines.
Mechanical: among the strongest materials known
Carbon nanotubes have tensile strengths reported between 11 and 100 gigapascals depending on diameter and defect content, with a Young’s modulus near 1 terapascal for SWCNT. For context: high-strength carbon steel reaches roughly 1.5 GPa tensile, and titanium alloys reach 1.4 GPa. At one-sixth the density of steel, the specific strength of an ideal carbon nanotube exceeds anything in conventional engineering materials.
The mechanical implications matter most in composites, where a small mass loading of carbon nanotubes (5–15 wt%) can substantially improve the strength, stiffness, and toughness of a polymer matrix. The challenge is dispersion — bundled or agglomerated CNTs deliver only a fraction of theoretical reinforcement.
Electrical: metallic conduction at the molecular scale
A single armchair SWCNT carries current at metallic densities along its axis without electron-phonon scattering for length scales up to several microns at room temperature. Current densities of 109 A/cm2 have been measured in individual SWCNT — roughly 1,000× the safe sustained current density in copper interconnects. MWCNT bundles in practical conductive films achieve 102–104 S/m bulk conductivity, far above any conventional carbon black at equivalent mass loading.
This is the property exploited in lithium-ion battery cathode conductive additives — replacing 2–5 wt% carbon black with 0.3–1.5 wt% MWCNT cuts the conductive-additive mass burden by 3–10× while improving rate capability through reduced junction resistance.
Thermal: axial conduction beyond copper
Theoretical axial thermal conductivity of an isolated SWCNT exceeds 3,000 W/m·K at room temperature — higher than copper (401 W/m·K) and approaching diamond (~2,000 W/m·K, isotropic). In practice, CNT mat or film thermal conductivity is reduced by inter-tube junction resistance and impurities, but optimized aligned-CNT structures achieve 100–500 W/m·K axially. This makes carbon nanotubes attractive for thermal interface materials, heat dissipation films, and emerging thermal management in electronics.
Surface area and one-dimensional geometry
Despite being closed cylinders, carbon nanotubes have measurable BET surface areas of 100–1,300 m²/g depending on bundling and end-cap state. More important than absolute surface area is the one-dimensional geometry: high aspect ratio (length-to-diameter ratio from 100 to over 10,000) gives carbon nanotubes a very low percolation threshold for conductive networks. A 0.05 vol% loading of a 1,000-aspect-ratio nanotube forms a connected network — versus 16 vol% for spherical particles of equivalent size.
This is why carbon nanotubes work as conductive additives at one-third to one-tenth the loading of carbon black, why they enable transparent conductive films, and why they appear as the active scaffolding in silicon-CNT lithium-ion anode composites and supercapacitor electrodes.
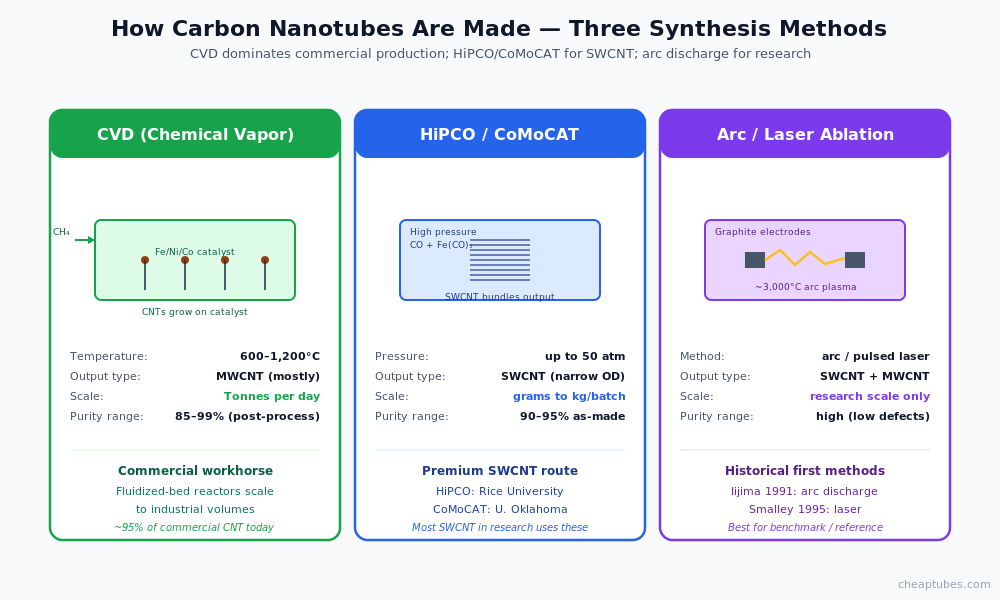
How carbon nanotubes are made
Three synthesis methods dominate the commercial and research landscape today.
CVD (chemical vapor deposition)
The dominant commercial route, accounting for the great majority of MWCNT tonnage produced today. A hydrocarbon feedstock (methane, ethylene, acetylene, or higher hydrocarbons) is decomposed catalytically over a metal catalyst (iron, nickel, cobalt, or molybdenum supported on alumina, silica, or magnesium oxide) at 600–1,200°C in a reactor under atmospheric or low pressure. The catalyst nanoparticle determines the resulting nanotube diameter, and process conditions control wall count, length, and defect density. CVD scales well — fluidized-bed reactors operating continuously produce tens to hundreds of kilograms per day per reactor at industrial sites. Output is MWCNT-dominant; SWCNT-targeted CVD is possible but yields are lower and post-processing more complex.
HiPCO and CoMoCAT for SWCNT
For SWCNT, two specialized CVD variants dominate. HiPCO (High-Pressure CO disproportionation, Rice University origin) uses carbon monoxide as the feedstock and iron pentacarbonyl as the catalyst precursor at pressures up to 50 atmospheres. CoMoCAT (Cobalt-Molybdenum catalyst, University of Oklahoma origin) uses a supported bimetallic catalyst at lower pressure. Both produce relatively narrow-distribution SWCNT but at smaller batch sizes than MWCNT CVD.
Arc discharge and laser ablation
The historical first-production methods. Arc discharge between graphite electrodes in inert atmosphere produces MWCNT (Iijima’s original method) and, with metal catalysts in the electrodes, SWCNT. Laser ablation of metal-doped graphite targets yields high-purity SWCNT in modest quantities. Both methods are limited to research scale today but produce highly graphitic, low-defect carbon nanotubes that remain useful for fundamental studies and benchmark characterization.
After synthesis, carbon nanotubes are typically post-processed: acid washing to remove residual metal catalyst, annealing under inert atmosphere to improve crystallinity, and optionally functionalized to introduce surface groups (-COOH, -OH, -NH2) for solvent compatibility.
Where carbon nanotubes are used
Carbon nanotube applications span academic research, pilot-scale manufacturing, and mature commercial production. Grouped by maturity:
Mature commercial applications (tonnage today)
- Lithium-ion battery cathode conductive additive — MWCNT 8–20 nm replacing carbon black in LFP and NMC cathodes. The largest CNT application by tonnage today.
- Polymer composites — automotive parts, sports equipment, electronics housings. MWCNT or functionalized variants typically loaded at 1–10 wt%.
- Conductive plastics — electrostatic-discharge-safe enclosures, fuel lines, semiconductor packaging.
- EMI shielding films and coatings — wrapping cables, lining cabinets, replacing metal foils in flexible applications.
- Conductive inks and coatings — printed electronics, transparent conductive films.
Pilot and growing commercial
- Silicon-CNT anode composites — buffering the 300% volume expansion of silicon in next-generation lithium-ion anodes. Production cells shipping since 2024.
- Supercapacitor electrodes — high-rate energy storage devices, often paired with graphene or activated carbon.
- Thermal interface materials — heat dissipation between high-power chips and heat sinks.
- Field-emission electron sources — flat-panel displays, electron microscopy, X-ray sources.
R&D and specialty
- Lithium-sulfur cathode scaffolds — porous CNT networks hosting sulfur in next-generation high-energy batteries.
- Drug delivery — functionalized SWCNT carrying chemotherapy agents into tumor cells.
- Single-electron transistors and quantum electronics — exploiting the bandgap of semiconducting SWCNT.
- Aerospace structural composites — carbon nanotube fibers and yarns for ultra-high-specific-strength structures.
- Water filtration and desalination — vertically aligned CNT forests as ultra-selective membranes.
Safety and handling considerations
Carbon nanotubes are a relatively new material class, and toxicology research is ongoing. The current consensus, based on inhalation studies in animal models and limited human-exposure cohorts, divides the safety profile by morphology:
- Long, rigid MWCNT (≥5 µm length, low flexibility) have shown asbestos-like inflammatory and fibrotic responses in animal lung-deposition studies. The U.S. National Institute for Occupational Safety and Health (NIOSH) classifies these in a precautionary risk category.
- Short MWCNT (≤5 µm length) and most SWCNT exhibit less concerning responses in the same studies, though the data are not as extensive.
- Functionalized CNT (especially carboxylated or hydroxylated variants) clear more rapidly from biological tissues than pristine CNT.
The NIOSH Recommended Exposure Limit (REL) for all carbon nanotubes is 1 µg/m³ (8-hour time-weighted average) — a precautionary value reflecting current uncertainty rather than a documented dose-response threshold. The inhalation route is the primary concern; dermal and oral exposure routes show much lower risk in the available data.
Standard handling practices for laboratory and production environments include: HEPA-filtered local exhaust ventilation, glove boxes or fume hoods for dry powder handling, surfactant-stabilized aqueous dispersions or pre-dispersed slurries for liquid-phase work (which dramatically reduce airborne exposure), and respiratory protection where engineering controls are insufficient. Cheap Tubes provides a Safety Data Sheet (SDS) for every catalog product.
How to choose the right carbon nanotube
The right carbon nanotube depends on the application. A decision tree:
- Cathode conductive additive for Li-ion → MWCNT 8–20 nm, 95–98% purity, 0.3–1.5 wt% loading. See the MWCNT Buying Guide.
- Silicon-CNT anode composite → MWCNT 98%+ or SWCNT 95–99%, 1–5 wt% loading. See the Silicon Anode Guide.
- Polymer composite at industrial scale → MWCNT masterbatch (pre-dispersed in PA6, PC, ABS, or PP) at 1–10 wt% loading. See CNT Masterbatches.
- EMI shielding or conductive coating → Functionalized MWCNT or pre-dispersed aqueous formulation. See Functionalized CNT.
- Academic R&D or publication-grade work → MWCNT 99.9% or SWCNT 99% with per-batch characterization data (TGA, BET, Raman, TEM). See the SWCNT Buying Guide.
- High-rate or premium battery cells → SWCNT 95–99% at 0.05–0.5 wt% loading. See SWCNT catalog.
For dispersion-limited applications at pilot or production scale, Flexiphene patented surfactant-stabilized dispersions (U.S. Patents 10,049,783 and 11,961,630) deliver stable carbon nanomaterial loadings in nylon, polyester, polyimide, polyamide, polyaminotriazole, and polyaniline polymer matrices — the binder systems the battery industry is migrating to.
Authoritative external references
- Iijima, S. — Helical microtubules of graphitic carbon (1991) (Nature 354, 56–58) — the discovery paper.
- Saito, R., Dresselhaus, G. & Dresselhaus, M.S. — Physical Properties of Carbon Nanotubes (Imperial College Press, 1998) — the foundational textbook.
- De Volder, M.F.L., Tawfick, S.H., Baughman, R.H. & Hart, A.J. — Carbon nanotubes: present and future commercial applications (2013) (Science 339, 535–539) — the modern commercial review.
- Tong, X. et al. — A review on applications and challenges of carbon nanotubes in lithium-ion battery (2025) (Carbon Energy) — comprehensive 2025 update on battery applications.
- NIOSH — Current Intelligence Bulletin 65: Occupational Exposure to Carbon Nanotubes and Nanofibers (2013) — exposure limits and handling guidance.
Frequently asked questions
What is the difference between SWCNT and MWCNT?
A single-walled carbon nanotube (SWCNT) is one cylindrical shell of graphene, typically 0.7–2 nanometers in outer diameter. A multi-walled carbon nanotube (MWCNT) is multiple concentric SWCNT-like shells nested together, typically 5–50 nanometers in outer diameter. SWCNT has higher theoretical mechanical strength and electrical conductivity per gram, while MWCNT is more affordable to produce, more mechanically robust to handle, and dominates industrial applications such as battery conductive additives and polymer composites.
Are carbon nanotubes safe?
Carbon nanotube safety depends on morphology. Long rigid multi-walled CNT (≥5 µm length) have shown inflammatory responses similar to asbestos fibers in animal inhalation studies and warrant precautionary handling. Short MWCNT, most SWCNT, and functionalized variants have shown less concerning profiles. The U.S. NIOSH Recommended Exposure Limit for all CNT is 1 µg/m³ as a precautionary value. Standard handling — HEPA ventilation, glove boxes for dry powder, surfactant-stabilized dispersions for wet-phase work — keeps occupational exposure well below this threshold.
Why are carbon nanotubes so expensive?
Single-walled carbon nanotube production is yield-limited: the synthesis conditions that produce SWCNT (HiPCO, CoMoCAT, laser ablation) operate at lower throughputs than the CVD processes that produce MWCNT, and downstream purification is more complex. The result is per-gram prices for SWCNT typically 5–20× higher than MWCNT. MWCNT itself is produced at tonnage scale and the per-kilogram price has fallen substantially since 2010. Production-grade MWCNT 95% purity is one of the more affordable engineering nanomaterials available.
What is the strongest carbon nanotube?
Defect-free single-walled carbon nanotubes have the highest measured tensile strength of any material — up to 100 gigapascals at the individual-tube level, compared to roughly 1–2 GPa for high-strength carbon steel. Practical strength in macroscopic CNT yarns or composites is much lower because of inter-tube slippage, defects, and matrix limitations, but specific strength (strength-to-density ratio) for SWCNT composites still exceeds anything in conventional engineering materials.
Can carbon nanotubes conduct electricity?
Yes. Metallic-chirality single-walled CNT and most multi-walled CNT conduct electricity along the tube axis at densities orders of magnitude beyond copper interconnects on a per-area basis. Semiconducting-chirality SWCNT has a tunable bandgap depending on tube diameter and is used in transistor and sensor applications. In a practical CNT network or composite, bulk electrical conductivity reaches 102–104 S/m — far above carbon black at equivalent mass loading.
What is the difference between carbon nanotubes and graphene?
Graphene is a one-atom-thick planar sheet of carbon atoms in a hexagonal lattice. A carbon nanotube is that same hexagonal sheet rolled into a seamless cylinder. The two materials share many physical properties (metallic-like conductivity in some configurations, high thermal conductivity, high mechanical strength), but the geometric difference — 2D sheet vs 1D cylinder — gives them different practical applications. Graphene excels at surface-coverage applications (films, coatings, supercapacitor electrodes). Carbon nanotubes excel at line-conduction and percolation applications (cathode additives, composites, sensors).
How are carbon nanotubes used in batteries?
Multi-walled carbon nanotubes are the dominant conductive additive in modern lithium-ion battery cathodes — replacing carbon black at one-third to one-tenth the loading thanks to their high aspect ratio. In silicon-carbon anode composites, MWCNT and graphene buffer the 300% volume expansion of silicon during lithiation while preserving electrical connectivity. In next-generation lithium-sulfur cells, porous CNT scaffolds host elemental sulfur and trap polysulfide intermediates that would otherwise destroy capacity. See the Graphene & CNT Battery Applications Hub for the full landscape.
Where can I buy research-grade carbon nanotubes?
Cheap Tubes Inc. has supplied research-grade carbon nanotubes for 21 years to academic researchers, R&D groups, and industrial customers. The catalog covers single-walled CNT (95–99.9% purity), multi-walled CNT (95–99.9% purity in 8–50 nm OD ranges), functionalized variants, masterbatches in engineering polymers, and pre-dispersed slurries. Per-batch characterization data (TGA, BET, Raman, TEM) is available on request. Browse the SWCNT catalog, MWCNT catalog, or contact Cheap Tubes Inc. for custom requirements.
Continue reading
- Single-Walled Carbon Nanotubes Buying Guide
- Multi-Walled Carbon Nanotubes Buying Guide
- Graphene Nanoplatelets Buying Guide
- Graphene & CNT Battery Applications Hub
- Carbon Nanotube Properties: Electrical, Mechanical & Thermal
- Carbon Nanotube Composites: Types, Properties & Applications
About the author
Mike Foley is the founder of Cheap Tubes Inc. and CTI Materials LLC. He holds two granted U.S. patents (10,049,783 and 11,961,630) in carbon nanomaterial applications, with additional patents in prosecution. His patented materials were selected by NASA for the Enceladus mission as a dual-capacitance layer in ion-selective electrodes. Mike has supplied carbon nanomaterials to academic research, industrial R&D, and battery production for 21 years.

